The Role of the Human Microbiome in Obesity-related Health
This activity expired October 1, 2019.
Course Overview: Recent studies suggest that maladapted gut microbiomes are attributed to a number of disease states, such as inflammatory bowel diseases, autoimmune diseases, cancer, diabetes, and obesity. This article highlights the relationship between the gut microbiome and the clinical implications it has, with an emphasis on the clinical sequalae surrounding obesity.
Course Description: This educational program is designed to educate, through independent study, multidisciplinary clinicians who care for the metabolic and bariatric surgery patient population.
Course Objectives: Upon completion of this program, the participant should be able to:Course Objectives: Upon completion of this program, the participant should be able to:
- Define the human microbiome.
- Discuss the role of the human microbiome.
- Describe the distribution of microbial organisms in the human body.
- List the beneficial effects of a healthy gut microbiota in humans.
- Discuss the role of probiotic supplementation and diet and their effect on gut microbiome.
- Explain the difference between prebiotic and probiotic.
Completion Time: This educational activity is accredited for a total of 1.5 contact hours.
Target Audience: This accredited program is intended for nurses who treat patients undergoing surgical treatment for obesity.
Provider: This educational program is provided by Matrix Medical Communications. Provider approved by the California Board of Registered Nursing, Provider Number 14887, for 1.5 contact hours.
Provider Contact Information: Emily A. Scullin Matrix Medical Communications, 1595 Paoli Pike, Suite 201, West Chester, PA 19380; E-mail: [email protected]

Bariatric Times. 2017;14(10):12–20
A Message from the Department Editor
Dear Colleagues:
It is with great pleasure that we present another wonderful contribution to our Integrated Health Continuing Education department on a stimulating topic that is one of the most exciting frontiers in medicine today—the role of the human microbiome in obesity-related health. The human gut harbors a complex community of microbes that profoundly influences many aspects of our health and well-being. Advances in this new frontier are very exciting because the gut appears to influence on many diseases commonly seen in patients with obesity. Heightened interest and research have expanded our understanding of gut health and its affect on our bodily systems. This new information allows the development of novel treatment strategies for many health disorders.
I hope you enjoy this in-depth review of the human gut microbiome. I believe further research into obesity and the gut microbiome will lead to the development of treatment modalities, that will alter clinical practice.
My best to you,
Tracy Martinez, RN, BSN, CBN
Tracy Martinez, RN, BSN, CBN, is Department Editor: Integrated Health Continuing Education, Bariatric Times; and Program Director, Wittgrove Bariatric Center, in La Jolla, California.
by Erik Sudin, BA, MD candidate and Fahad Zubair, MD
Erik Sudin, BA, is an MD candidate at Frank H. Netter MD School of Medicine at Quinnipiac University in North Haven, Connecticut. Fahad Zabair, MD, is with the Geisinger Medical Center in Danville, Pennsylvania.
Funding: Support for this activity was provided by Bariatric Advantage
Disclosures: The author reports no conflicts of interest relevant to the content of this article.
Abstract: The term human microbiome refers to the total genetic material of all organisms colonizing a human being. Because the gut is the most heavily colonized area of the human body, the term microbiome is often used to refer to the genomes of all the microbes populating a given individual’s gastrointestinal tract. The microbiome of one person can vary dramatically from that of another person, and the clinical consequences of that variation can have profound impacts on a person’s health, including a pivotal role in his or her obesity status. Ongoing research continues to shed light on the complex interactions between human hosts and their microbiomes and how the microbiome can affect overall health of a given individual. Recent studies suggest that maladapted gut microbiomes are attributed to a number of disease states, including inflammatory bowel diseases, autoimmune diseases, cancer, diabetes, and obesity. This article highlights the relationship between the gut microbiome and the clinical sequalae surrounding obesity.
Keywords: Obesity, human microbiome, gut microbiome, microbiota, digestion, microbiology, genomics, short-chain fatty acids, oxalate, carcinoma, bariatric surgery, fecal transplants, probiotics, prebiotics
Introduction
Within every human being exists an entire ecosystem of microorganisms.1 Microbial cells of the human body are referred to collectively as the microbiota. The collective genomes of the microbes that live within and on the human body are termed as the human microbiome, which includes bacteria, fungi, protists, archaea, and viruses, such as bacteriophages (i.e., viruses that infect bacteria [Table 1].[1,2]

Though research into this complex system is ongoing, the current body of literature continues to unfold an exciting narrative full of clinical potential, showing just how the microbiome functions throughout human development and to adulthood, in both healthy and ill individuals. In particular, there is a growing area of research devoted to the gut microbiome’s role in diseases, such as inflammatory bowel diseases, autoimmune diseases, cancer, diabetes, and obesity.
The microbiota surpasses human cells in the quantity of genes within their genomes, and contains, in orders of magnitude, more genes than are present in their human hosts.[3] Since the completion of the Human Genome Project, the number of genes in the human genome has been revised several times but is currently estimated to be in the tens of thousands, with as many as 19,000 gene coding for proteins.[4] This number is dramatically overshadowed by the microbial genomes contributing to the microbiome, which is now believed to contain nearly 10 million genes just within the intestinal region of the gastrointestinal tract alone.[5]
The microbiota becomes a part of us before we are born and remains a part of us throughout our entire life. Studies examining the meconium of healthy neonates suggest that humans are able to populate their microflora via mother-to-child efflux of commensal bacteria in utero.[6] This population of microflora changes dramatically during early childhood development, depending on our diet, genetics, and other environmental factors, eventually establishing a degree of stabilization in composition by the age of five years. However, this microbial ecosystem is still dynamic during adulthood, remaining ever susceptible to changes in diet, disease, and clinical intervention.[7]
The Human Microbiome Project (HMP) is an initiative that was launched in 2008 by the United States National Institutes of Health. The goal of the HMP was to identify and characterize the microorganisms found in both healthy and diseased humans. Figure 1 shows the distribution of sequenced microbial organisms with respect to their host anatomy. Note that more than half of the representation exists in the oral cavity and gastrointestinal tract. Furthermore, the project aimed to understand the microbial components of the human genetic and metabolic landscape, particularly to illuminate how these microbial components contribute to our normal human physiology and disease predisposition.[3]
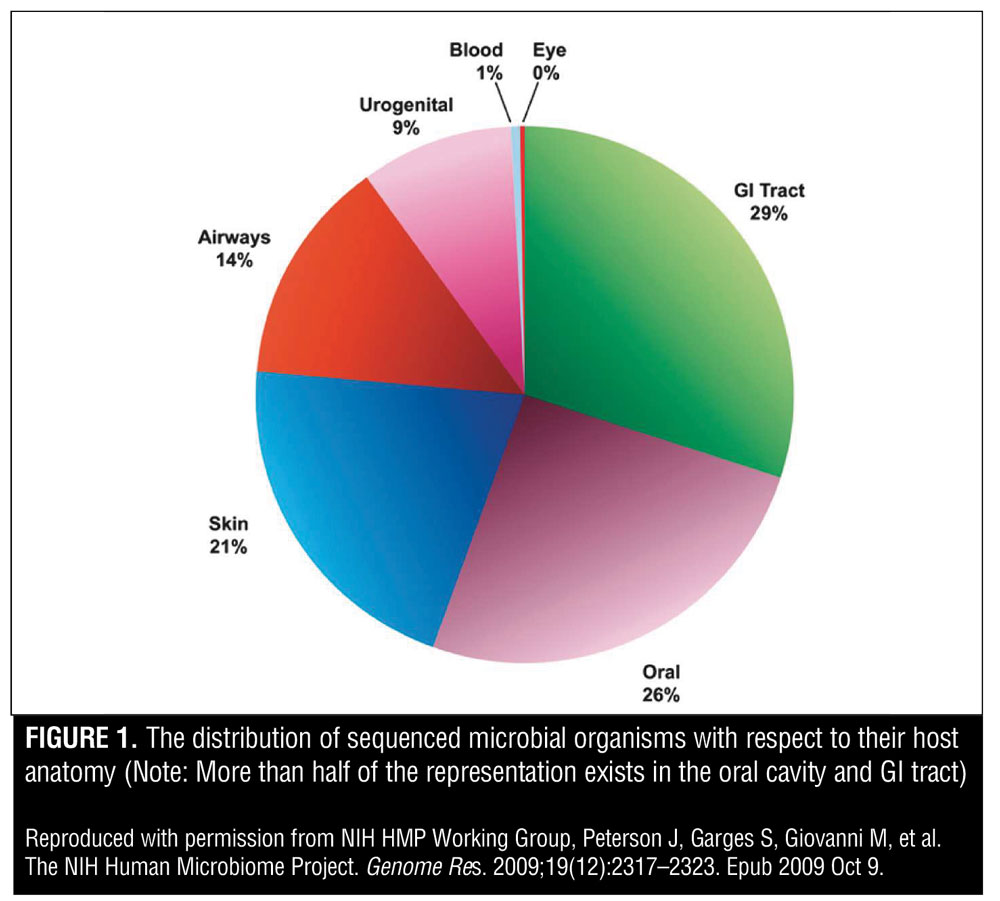
While laying the groundwork for characterizing the microbial landscape and its relationship to humans, the HMP introduced the concept of a “human supraorganism” featuring two distinct characteristics. First, the human supraorganism is a composite of microbial and human species. Second, it is a summation of the genes embedded in our human genome and microbiome. This novel concept proposes that our metabolic system features a coalescence of human and microbial traits.[1,2]
Hosting more than 100 trillion microbial cells of the microbiota, the gut quickly became a research focus of the HMP.8 With so many varied genetic and enzymatic processes at play within a single gut, it is no surprise that the microbiome was found to play an important role in a variety of clinical presentations. In particular, a given individual’s specific variations of the microbiota have shown to play pivotal roles in behavior, metabolism, and obesity.
The Gut Microbiome and Overall Health
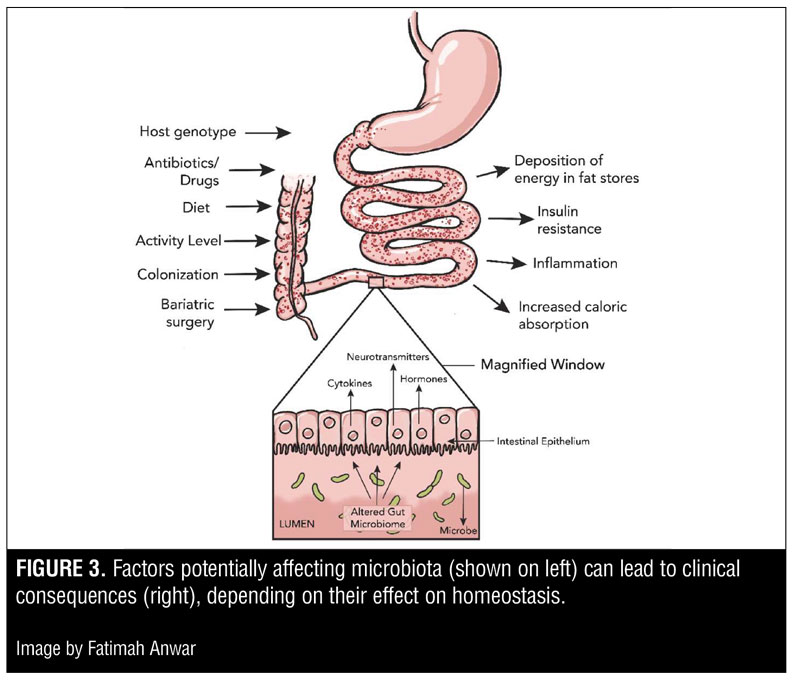
Microbiomics, the study of the microbiome, continues to largely focus on the distal gut, i.e., that part of the gastrointestinal tract that contains the greatest number of microbes.[9,10] The distal gut is where the body synthesizes essential amino acids and vitamins and processes components of otherwise indigestible contributions to our diet, such as plant polysaccharides. The microbial communities, and hence the contributing microbiomes of the distal gut, vary significantly among all human beings. The factors influencing the profile of microbiota in the gut include ecological interactions within hosts, variations in genotype, ingestion of drugs, colonization history, exercise, and diet.[11–13] Substantial variation is also present in a variety of disease states, and it is now understood that an altered microbiome may not only be a result of disease, but also a cause of it.[12]
The gut microbiota has many beneficial effects, including development and homeostasis of the immune system, support of food digestion and fat metabolism, maintainence of epithelial homeostasis, promotion of angiogenesis, regulation of the enteric nervous system, and prevention of colonization by pathogens.[14,15] Conversely, maladapted or altered gut microbiota might affect these processes adversely, giving rise to a number of disease states. In mammals, an altered gut microbiome may impact a variety of processes, including gastrointestinal health, nutritional status, drug metabolism, predispositions for carcinoma, immune response, and psychology.[16]
Detoxification and cancer. The human microbiome is, by many measures, a chemical factory. As we will see with respect to obesity, it plays a myriad of roles. In addition to nutritionally consequential metabolic processes, the human microbiome plays a role in other critical processes of the gastrointestinal tract. In particular, there is a growing area of research devoted to the exploration of the microbiota and its role in drug metabolism, detoxification, and, increasingly, in the onset of cancer.
Oxalic acid homeostasis in the human body is an example of a physiological process that is dependent on the detoxifying characteristics of the microbiota. Metabolism of oxalate by the microbiota has been shown in rats to have a relationship to the formation of renal stones.[2] More specifically, there is a bacterium of the microbiota called Oxalobacter formigenes, which is found to be a negative risk factor for calcium oxalate kidney stone disease. Sidhu et al[17] demonstrated that by degrading dietary oxalate, this bacterium contributes to oxalic acid homeostasis in its mammalian host. Discussions have already begun on how discoveries such as these might lead to probiotic use to prevent clinical consequences due to urinary oxalate, and ultimately to reduce risk of kidney stones.
While oxalate is just one example of a substance on which the microbiota act to detoxify the human host, a variety of other molecules are also affected. Carcinogenesis can be affected by multiple physiological reactions and molecular influences. By contributing to the chemical environment of gut stem cells, promoting inflammatory responses, and producing variable amounts of butyrate—known to affect deoxyribonucleic acid (DNA) integrity—the microbiota continue to be an area of research exploring how they can be manipulated to influence disease course.[18]
Helicobacter pylori (H. pylori) is a bacterium often found in patients with gastric ulcers. Through the examination of specific genetic markers of H. pylori, and an understanding of how it causes gastric ulcers, researchers have now also implicated it as a determinant of gastrointestinal cancer. Abreu et al[18] examined the genetic profile of H. pylori with respect to a gene called cytotoxin-associate gene (Cag). When H. pylori was positive for Cag, it was found to contribute significantly to distal gastric cancer. While the genetic profiling showing the role of Cag in carcinogenesis is just one of many specific examples, it illuminates an area of research that is rapidly advancing. In the case of Cag, the mechanism of the protein interaction between Cag- H. pylori and Cag+ H. pylori has been shown to be the pivotal protein interaction that incites malignancy among surrounding human cells.[18]
Immune response disruption. H. Pylori is a clinically familiar example of a microbiotic factor that can cause chronic inflammation in the gut, but less well-known examples continue to enter the arena of research on inflammation with respect to the human microbiome.[19] The activity of the microbiota with respect to innate and adaptive immune response is now thought to have far-reaching consequences on diseases such as asthma, ulcerative colitis, and Crohns’ disease, a disease characterized by a chronic inflammatory state associated with endothelial dysfunction and a “leaky gut.”[2,20–23]
Atopy is generally defined as a genetic tendency toward developing allergic diseases, such as rhinitis, asthma, and atopic dermatitis (also known as eczema).[24] It is typically characterized by a hyperactivation of T helper 2 cells and associated immunological pathways, including particularly elevated serum IgE levels. It might seem odd that the microbiota, often associated with the human gastrointestinal tract, could play a role in respiratory diseases such as asthma or atopy; however, through the longitudinal examination of neonatal gastrointestinal tracts through development, Fujimura et al[20] observed that children with microbiomes enriched with Escherichia coli or Clostridium difficile were at a significantly higher risk for developing atopy.
It has been long known that a patient’s obesity status can play a role in inflammatory responses. While the exact relationship between obesity, inflammation, and diabetes is not entirely clear, studies suggest that inflammatory cytokine activity can predict weight gain, and laboratory manipulation of inflammatory cytokine levels in mouse models can advance insulin resistance.[25] Immunological effects of the microbiota are widespread enough to contribute to inflammatory and respiratory disease, which indicates that further study of the relationship between obesity, inflammation, and the human microbiome is warranted.
Other altered processes. As research continues, there are a multitude of other physiological processes that appear to be affected by the microbiome. Alterations in the microbiome appear to affect enterohepatic circulation of bile, resulting in biliary disease.[26] Slower epithelial cell renewal results in decreased capacity to repair mucosal damage, which can also be altered by variations of the microbiome.[27] Decreased capacity to resist colonization with pathogens and a consequent increased risk for C. diff infection has been linked to differences among patient microbiomes.[2] As the potential clinical applications of microflora continue to be discovered, there is increasing evidence that mood (e.g., depression, anxiety) is also affected by the microbiome. As research continues to grow, the role of the human microbiome continues to shed light on clinical conditions once thought to act independently of gut microflora.
The Gut Microbiome and Obesity
The relationship between the human microbiome and the human genome continues to demonstrate a complex interplay contributing to the overall metabolism of the aforementioned “human supraorganism.” As such, scientists that hitherto relied on human genomes to look for factors contributing to disease have begun examining the genomes of the microflora inhabiting the human gastrointestinal tract. This area of research continues to make use of molecular tools to unwind the intricate relationship that contributes to overall human nutrition and obesity status.
With the advancements of genetic sequencing, scientists have had the opportunity to explore the expression of proteins that have historically been proven critical to metabolic processes of the gastrointestinal tract. Through microbiome sequencing, Gill et al[9] quantifiably examined the expression of nutrition-related proteins within the human microbiota. While complex to decipher, the differential expression they examined within the microbiota ultimately suggests that variations in metabolic processes among different individuals might depend necessarily on the microbiome. Depending on the expression patterns of the microbiota, processing of nutrients via known metabolic processes consequently varies between different patients. This genetic profiling of microbiomes would suggest a link between the microbiome and metabolic activities seen in nutritional deficiencies as well as obesity.[9]
Metagenomic studies have also demonstrated that the differential genetic expression of certain genes relating to obesity within a mouse host model relates directly to the functional role of the mouse microbiota. In particular, Turnbaugh et al[10] designed a study showing that “obese-type” mice, homozygous for an obesity mutation (ob/ob), exhibit different microbiotic abilities to digest polysaccharides of plants into short-chain fatty acids. These short-chain fatty acids might then contribute an individual mouse’s caloric intake. Furthermore, when the microbiota of obese (ob/ob) mice were transplanted into germ-free adult wild-type mice (+/+), a degree of fat deposition was shown to increase at a greater rate than in the controls of transplanted microbiota from other wild-type mice.[2,10]
The studies relating the microbial gut ecosystem of obesity are not limited strictly to rodents. Riduara et al[28] examined pairs of human twins with discordant body mass, and transplanted their fecal material into germ-free (GF) mice of normal weight. The mice colonized with fecal microbiota from the twin with obesity developed obesity, whereas the mice colonized with fecal microbiota from the lean twin remained lean. Differences in body composition were attributed to increases in the following: 1) fermentation of short-chain fatty acids in the lean mice; 2) microbial transformation of bile acids in the lean mice; and 3) metabolism of branched-chain amino acids in the obese mice. The lean and obese mice were then housed together. It was discovered that gradually the obese mice became lean and their gut flora began to resemble that of the lean mice. However, the reverse was not true, as the lean mice resisted invasion from obese mouse gut flora.[28]
As discussed, there are several nutritional and immunological relationships affecting the complex disease state we call obesity. Notably, microbial cells of the gut allow humans to harvest otherwise inaccessible nutrients from their diet by converting indigestible plant polysaccharides into short-chain fatty acids.[9,10,29] However, this is just one example of many metabolic effectors we have yet to understand, and changes to the microbiota might also affect obesity via insulin-sensitivity, which itself can contribute to changes in metabolic disease.[25] Even as the study of the microbiome advances, many of these relationships are still unclear.
Gut microbes impact host metabolism via generation of various proteins, including hormones and neurotransmitters that can activate signaling pathways in the gut and also enter the circulation, resulting in effects on insulin resistance, inflammation, and deposition of energy in fat stores (Figure 2).[30,31] Furthermore, the aforementioned microbial production of short-chain fatty acids that resulted in adipose deposition have a second clinical role. They also affect anti-inflammatory pathways and glucose metabolism by acting as signaling molecules that affect immune cell production of chemokines and interleukins in the gastrointestinal tract.[32–34]
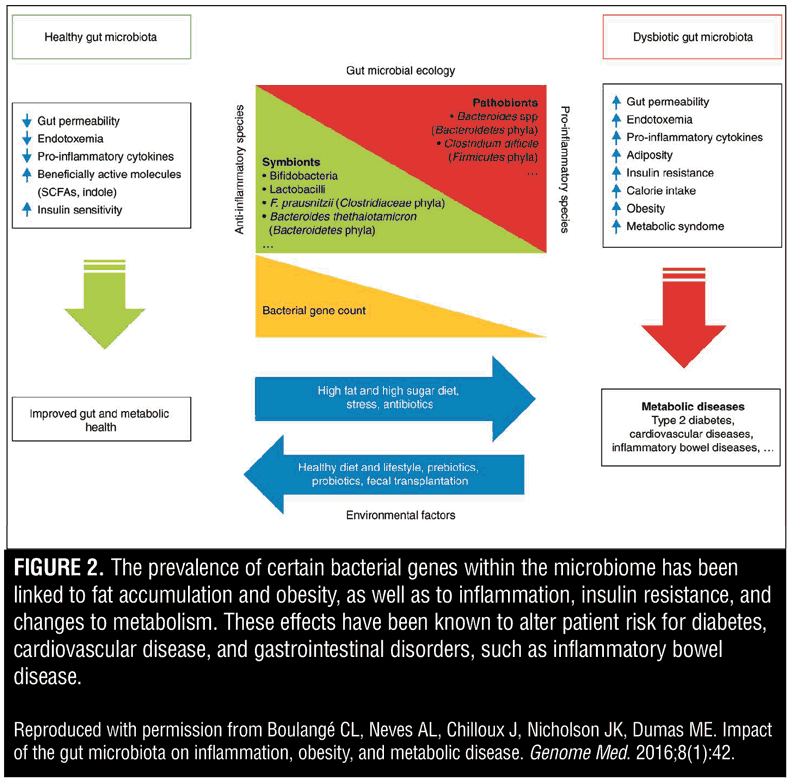
Various studies conducted in mouse models have examined the role of gut microbiota in insulin resistance, inflammation, energy intake, absorption, and storage—all potential factors that contribute to obesity and type 2 diabetes mellitus (T2DM).
Bäckhed et al[35] hypothesized that the microbiota act through an integrated host signaling pathway to regulate energy storage in the host. To test this idea, the researchers colonized adult germ-free (GF) lean mice, raised in the absence of microorganisms, with the gut microbiota from conventionally raised animals that harbored a microbiota beginning at birth. The authors found that, compared to the GF group, the conventionally raised mice displayed a 60-percent increase in body fat content and relative insulin resistance within 14 days despite reduced food intake.
Additional studies from Bäckhed et al revealed that when GF mice and mice colonized with gut microbiota from conventionally raised animals were fed a diet high in fat and carbohydrates, the colonized mice gained significantly more weight than the GF mice. In fact, weight gain among the GF mice consuming a high-fat, high-carbohydrate diet was no different when compared to GF mice on a low-fat diet. This finding illustrates that the GF mice were protected against obesity produced by consumption of a high-fat, high-carbohydrate diet. The proposed mechanism of this protection is two-fold: 1) the GF mice expressed increased levels of phosphorylated AMP-activated protein kinase in muscle and liver, which stimulated fatty acid oxidation in peripheral tissues; and 2) the GF mice had increased intestinal fasting-induced adipose factor.[36]
Microbial change after bariatric surgery. Bariatric surgery is a complex clinical pursuit that relies heavily on the expertise and well-informed decisions of highly skilled physicians. With such dramatic discoveries taking place in microbiomics, and with variations between individual patients being studied, it is no surprise that bariatric surgery—a field predicated on carefully planned clinical procedures leading to life changing homeostatic alterations—is an area of medicine that can benefit from the development and use of clinical tools that take advantage of certain changes to the gut microbiota.
The impact that the human microbiome has on gastric bypass surgery is a growing area of research. In an effort to determine whether the mechanism underlying weight loss after Roux-en-Y gastric bypass (RYGB) is in part due to subsequent changes in an individual’s gut microbiome after surgery, Liou et al[37] transferred gut microbiota from RYGB-treated mice to lean germ-free mice. Another group of lean germ-free mice received the gut microbiome from lean mice who underwent a sham surgery. After two weeks, the RYGB-transplanted mice lost weight and their fat mass decreased significantly compared to the sham surgery-transplanted mice, potentially due to altered microbial production of short-chain fatty acids.[37]
This research supports that there are transmissible and modifiable interactions between diet and gut microbiota. Furthermore, the resulting microbial communities in the gut can modulate host phenotype and adiposity levels.
Restoring the Gut Microbiome
The gut microbiome composition is strongly affected by dietary patterns. Turnbaugh and Ley[28,29] demonstrated that a high-fat and high-sugar diet increases the relative abundance of Firmicutes at the expense of the Bacteroidetes, both phyla of bacteria largely constituting the gut microbiome. Further, a low-calorie diet was implicated as potentially increasing the relative abundance of Bacteroidetes, gram-negative, nonsporeforming, anaerobic or aerobic bacteria, in individuals with obesity.[38,39]
Firmicutes is another phylum of bacteria in the gut. The ratio of Firmicutes to Bacteroidetes in the gut is complex and has revealed important correlations with respect to obesity status. At first glance, it would seem that a shift toward a ratio that favors Bacteroidetes over Firmicutes would be beneficial. However, while some studies reveal important correlations with seemingly direct practical applications, there are also discussions on how modifying the biodiversity of the microbiome might yet transcend obesity research and have other clinical consequences to patient health.[40,41]
While microbiomic considerations with respect to obesity treatment seem difficult to navigate, it is important to note that with the current ongoing research our understanding of the complex microbiotic human ecosystem, continues to expand.
It might make sense to replenish the microbiota with bacteria equipped to perform enzymatic activities to promote ideal health. Prebiotics and probiotics are two of the mechanisms that influence the clinical presentations related to the human microbiome.
If we, as clinicians, could control the proportion of bacteria in the gut that convert polysaccharides to short-chain fatty acids, control the microbial cells that contribute to atopy, or even control the microbiotic balance of oxalic acid metabolism, we could change the homeostatic equilibrium to favor patient health. Ongoing scientific research of the gut microbiome and its contributions to our health has helped scientists understand the relationship and mechanisms of action in both lean and obese models. Through this research, the state of the gut microbiome has emerged as an essential target in weight loss management. Intentional alteration of the microbial communities might be helpful in restoring nutritional balance for individuals with obesity.[2,10,42]
Monitoring the human microbiome will become increasingly important as clinical tools to alter the microbiome progress. The gut microbiome is tested by obtaining a stool culture. Individuals with obesity typically display a decrease of Bacteroidetes and increase in Firmicutes, whereas the opposite is expected in lean individuals.[36]
Research on treatment aimed at changing the gut ecosystem is promising. Modulating the ratio of Bacteroidetes to Firmicutes is an example of a clinical pursuit that might benefit patients. Restoring good balance to the gut microbiome in individuals who demonstrate an imbalance has become an emerging target of therapeutic intervention across multiple conditions, many of which are still being explored with respect to their microbiomic relationships.[36]
Probiotics. As our understanding of the microbiome advances, guidelines for clinical intervention via dietary changes must be optimized to improve patient health. Probiotics are often at the forefront of this discussion. Generally, probiotics are defined as live microorganisms, such as those seen in our microbiota, that are believed to improve the health of the human host that consumes them (Table 2).[43]

While the mechanisms of action can vary from enzymatic processes to yet uncharted health-promoting biochemical processes, the use of probiotics to improve health is not a new concept. Yogurt, for example, is often advertised as containing certain bacteria (probiotics) to help maintain a healthy microflora in the gut and improve digestive health.
More recently, less conventional means of introducing probiotics have become prevalent.[44] The use of fecal transplants to improve health has become a particular area of interest in large part for treating C. diff infection.[44,45] By using a fecal transplant, the fine balance of microbial activities in the gut are not fully deciphered and modulated, but rather one microbial ecosystem is replaced or complemented by another to improve the probability of a healthy gut microbiota. As research continues, the fine-tuning modulation of microflora might yet replace the use of fecal transplants.
Prebiotics. While the term probiotic is familiar to most people, prebiotics also play a novel role in treatment of obesity and other diseases by working carefully with the microbiota of patients. Prebiotics are not microbiota at all, but rather food substances that can be consumed to promote the growth of beneficial gut microflora (Table 2).[43]
Those with a flourishing microbiota in their guts likely consume prebiotics already; however, the traditional view of obesity treatment hinges on nutrition from the standpoint of calories, fat, protein, carbohydrates, fiber, and vitamins. The ratio of these nutritional components is critical, especially postoperatively for bariatric patients, in accordance with laboratory test results and other clinical guidelines. However, the idea of using prebiotics might alter the perspective of food digestion and absorption to include understand which microorganisms flourish best with which foods. By some measures, a prebiotic agent (often a nondigestible oligosaccharide) could be thought of as a “fertilizer” that enhances the growth of more beneficial species in the gut microbiome. As research into the role of prebiotics advances, the goal of nutritionists might become to cater to the biochemical “appetite” of gut microflora, in addition to the utilization of traditional nutritional guidelines.[32,43]
Conclusion
As the obesity epidemic continues to affect an increasing number of people around the world, researchers and clinicians continue to seek solutions. A once simplified physiological equation, we now know that the disease state known as obesity hinges on a variety of factors, including socioeconomics, psychology, cultural backgrounds, genetics, and endocrinology. With technological advances such as metagenomic sequencing, scientists are now exploring the once overlooked gut microbiome and the key role that it appears to play in our health. Studies in animal models have uncovered much about the complex system of the gut microbiome. More human studies and randomized, controlled trials will help further outline the manipulation of gut microbiota as a therapeutic measure in the management of obesity. Modification of the gut microbiome in an effort to restore healthy microflora with the use of antibiotics, probiotics, and prebiotics may be crucial to help patients with obesity lose a significant amount of weight and sustain a healthy lifestyle. Certainly, the identification and thorough understanding of the relationship between obesity and the gut microbiota are essential for establishing optimal weight management guidelines and treatments for obesity in a modern world.
References
- Peterson J, Garges S, Giovanni M, et al. The NIH human microbiome project. Genome Res. 2009;19(12):2317–2323.
- Turnbaugh PJ, Ley RE, Hamady M, et al. The human microbiome project: exploring the microbial part of ourselves in a changing world. Nature. 2007;449(7164):804–810.
- NIH Human Microbiome Project. Overview https://www.hmpdacc.org/hmp/overview/. Accessed August 8, 2017.
- Ezkurdia I, Juan D, Rodriguez JM, et al. Multiple evidence strands suggest that there may be as few as 19,000 human protein-coding genes. Hum Mol Genet. 2014;23(22):5866–5878
- Dietert RR, Dietert JM. The microbiome and sustainable healthcare. Healthcare (Basel). 2015;3(1):100–129.
- Jiménez E, Marín ML, Martín R, et al. Is meconium from healthy newborns actually sterile?. Res Microbiol. 2008;159(3):187–193
- Rodríguez JM, Murphy K, Stanton C, et al. The composition of the gut microbiota throughout life, with an emphasis on early life. Microb Ecol Health Dis. 2015;26:26050.
- Boulangé CL, Neves AL, Chilloux J, Nicholson JK, Dumas ME. Impact of the gut microbiota on inflammation, obesity, and metabolic disease. Genome Med. 2016;8(1):42.
- Gill SR, Pop M, Deboy RT, et al, Metagenomic analysis of the human distal gut microbiome. Science. 2006;312(5778):1355–1359.
- Turnbaugh PJ, Ley RE, Mahowald MA, et al. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature. 2006;444(7122):1027–1031.
- Dethlefsen L, McFall-Ngai M, Relman DA. An ecological and evolutionary perspective on human–microbe mutualism and disease. Nature. 2007;449(7164):811–818.
- Human Microbiome Project Consortium. Structure, function and diversity of the healthy human microbiome. Nature. 2012;486(7402):207–214.
- Clarke SF, Murphy EF, O’Sullivan O, et al. Exercise and associated dietary extremes impact on gut microbial diversity. Gut. 2014;63(12):1913–1920.
- Holmes E, Li JV, Athanasiou T, Ashrafian H, Nicholson JK. Understanding the role of gut microbiome-host metabolic signal disruption in health and disease. Trends Microbiol. 2011;19(7):349–359.
- Vollaard EJ, Clasener HA. Colonization resistance. Antimicrob Agents Chemother. 1994;38(3):409–414.
- Bianconi E, Piovesan A, Facchin F, et al. An estimation of the number of cells in the human body. Ann Hum Biol. 2013;40(6):463–471.
- Sidhu H, Allison MJ, Chow JM, Clark A, Peck AB. Rapid reversal of hyperoxaluria in a rat model after probiotic administration of Oxalobacter formigenes. J Urol. 2001;166(4):1487–1491.
- Abreu M, Peek RM Jr. Gastrointestinal malignancy and the microbiome. Gastroenterology. 2014;146(6):1534–1546.e3.
- Davis CP, Marks JW. H. pylori (Helicobacter Pylori) Infection. http://www.medicinenet.com/helicobacter_pylori/article.htm Accessed August 8, 2017
- Fujimura KE, Lynch SV. Microbiota in allergy and asthma and the emerging relationship with the gut microbiome. Cell Host Microbe. 2015;17(5):592–602.
- Greenblum, S, Turnbaugh PJ, Borenstein E, Metagenomic systems biology of the human gut microbiome reveals topological shifts associated with obesity and inflammatory bowel disease. Proc Natl Acad Sci U S A. 2012;109(2):594–599.
- Wellcome Trust Case Control Consortium. Genome-wide association study of 14,000 cases of seven common diseases and 3,000 shared controls. Nature. 2007;447(7145):661–678.
- Singh RK, Chang HW2, Yan D, et al. Influence of diet on the gut microbiome and implications for human health. J Transl Med. 2017;15(1):73.
- American Academy of Allergy, Asthma, and Immunology. Conditions Dictionary: Atopy. https://www.aaaai.org/conditions-and-treatments/conditions-dictionary/atopy. Accessed August 8, 2017.
- Yu C, Chen Y, Cline GW, et al. Mechanism by which fatty acids inhibit insulin activation of insulin receptor substrate-1 (IRS-1)-associated phosphatidylinositol 3-kinase activity in muscle. J Biol Chem. 2002;277(52):50230–50236.
- Kinross JM, Darzi AW, Nicholson JK. Gut microbiome-host interactions in health and disease. Genome Med. 2011;3(3):14.
- Pull SL, Doherty JM, Mills JC, Gordon JI, Stappenbeck TS. Activated macrophages are an adaptive element of the colonic epithelial progenitor niche necessary for regenerative responses to injury. Proc Natl Acad Sci U S A. 2005;102(1):99–104.
- Ridaura VK, Faith JJ, Rey FE, et al. Gut microbiota from twins discordant for obesity modulate metabolism in mice. Science. 2013;341(6150):1241214.
- Bäckhed F, Ley RE, Sonnenburg JL, Peterson DA, Gordon JI. Host-bacterial mutualism in the human intestine. Science. 2005;307(5717):1915–1920.
- Komaroff AL. The microbiome and risk for obesity and diabetes. JAMA. 2017;317(4):355–356.
- Ley RE. Obesity and the human microbiome. Curr Opin Gastroenterol. 2010;26(1):5–11.
- Gibson GR, Probert HM, Loo JV, Rastall RA, Roberfroid MB. Dietary modulation of the human colonic microbiota: updating the concept of prebiotics. Nutr Res Rev. 2004;17(2):259–275.
- Tazoe H1, Otomo Y, Karaki S, et al. Expression of short-chain fatty acid receptor GPR41 in the human colon. Biomed Res. 2009;30(3):149–156.
- Karaki S, Tazoe H, Hayashi H, et al. Expression of the short-chain fatty acid receptor, GPR43, in the human colon. J Mol Histol. 2008;39(2):135–142.
- Bäckhed F, Ding H, Wang T, et al. The gut microbiota as an environmental factor that regulates fat storage. Proc Natl Acad Sci U S A. 2004;101(44):15718-23. Epub 2004 Oct 25.
- Bäckhed F, Manchester JK, Semenkovich CF, Gordon JI. Mechanisms underlying the resistance to diet-induced obesity in germ-free mice. Proc Natl Acad Sci U S A. 2007;104(3):979–984.
- Liou AP, Paziuk M, Luevano JM Jr, et al. Conserved shifts in the gut microbiota due to gastric bypass reduce host weight and adiposity. Sci Transl Med. 2013;5(178):178ra41.
- Turnbaugh PJ, Bäckhed F, Fulton L, Gordon JI. Diet-induced obesity is linked to marked but reversible alterations in the mouse distal gut microbiome. Cell Host Microbe. 2008;3(4):213–223.
- Ley RE, Turnbaugh PJ, Klein S, Gordon JI. Microbial ecology: human gut microbes associated with obesity. Nature. 2006;444(7122):1022–1023.
- Chakraborti CK. New-found link between microbiota and obesity. World J Gastrointest Pathophysiol. 2015;6(4):110–119.
- Marchesi JR, Adams DH, Fava F, et al. The gut microbiota and host health: a new clinical frontier. Gut. 2016 Feb;65(2):330–339.
- Ley RE, Bäckhed F, Turnbaugh P, et al. Obesity alters gut microbial ecology. Proc Natl Acad Sci U S A. 2005;102(31):11070–11075.
- Feldman M, Friedman LS, Brandt LJ. Sleisenger and Fordtran’s Gastrointestinal and Liver Disease, Pathophysiology, Diagnosis, Management. Elsevier Health Sciences; 2015.
- Fuentes S, De vos WM. How to manipulate the microbiota: fecal microbiota transplantation. Adv Exp Med Biol. 2016;902:143–153.
- Liubakka A, Vaughn BP. Clostridium difficile Infection and fecal microbiota transplant. AACN Adv Crit Care. 2016;27(3):324–337.
Category: Past Articles







