Diabesity: The Worldwide Twin Epidemics of Obesity and Diabetes
This activity expired September 1, 2020.
Course Overview: Type 2 diabetes mellitus (T2DM) is a major public health problem that is significantly linked to another worldwide epidemic—obesity. Over the decades, as metabolic syndrome and metabolic surgery began rising to the forefront, the words “diabetes” and “obesity” were eventually unified to create the new, more descriptive term—diabesity. The authors provide statistics and predictions for both diseases, present management options for specific patient populations, describe multiple theories on why T2DM improves or, in some cases, resolves in individuals with obesity following metabolic surgery, and outline the latest consensus guidelines for T2DM treatment based on the available evidence.
Course Description: This educational program is designed to educate, through independent study, multidisciplinary clinicians who care for patients with overweight or obesity and T2DM.
Target Audience: This accredited program is intended for nurses who treat patients with overweight or obesity.
Completion Time: This educational activity is accredited for a total of 1.0 contact hour.
Provider: This educational program is provided by Matrix Medical Communications. Provider approved by the California Board of Registered Nursing, Provider Number 14887, for 1.0 contact hour.
Provider Contact Information: Emily A. Scullin Matrix Medical Communications, 1595 Paoli Pike, Suite 201, West Chester, PA 19380; E-mail: escullin@matrixmedcom.com
Funding: Support for this educational activity was provided by Medtronic, Minneapolis, Minnesota
 A Message from the Department Editor
A Message from the Department Editor
Dear Colleagues,
I am privileged to introduce an outstanding article about adiposity-based chronic disease and diabesity. You will learn the origin of two terms: diabesity and adiposity-based chronic disease or “ABCD.” This article also provides a through overview of the significance of diabesity and its medical and surgical treatment options, highlighting the impact these treatment options have on health risk reduction. Diabetes is a worldwide epidemic with severe health consequences and staggering cost.
We are fortunate to work in a field of medicine that can intervene and change the trajectory of the progression of such an expensive and potentially debilitating disease. We must work collaboratively to overcome the barriers to treatment that many patients face and be ambassadors for bariatric surgery as a treatment option for patients suffering from diabesity.
Lastly, I want to extend my gratitude to Drs. Higa, Shuchleib-Cung, and Akusoba for sharing their knowledge and expertise and supporting our continuing education series.
We have more wonderful articles planned for our Integrated Health Continuing Education series, and I look forward to presenting them to you.
My Best to You,
Tracy Martinez RN, BSN, CBN
Tracy Martinez, RN, BSN, CBN, is Department Editor: Integrated Health Continuing Education, Bariatric Times; and Program Director, Wittgrove Bariatric Center, in La Jolla, California.
by Ariel Shuchleib-Cung, MD; Ikemefuna Akusoba, MD; and Kelvin Higa, MD, FACS, FASMBS
Ariel Shuchleib-Cung, MD, and Ikemefuna Akusoba, MD, are Minimally Invasive Surgery Fellows at The University of California, San Francisco Fresno in Fresno, California. Kelvin Higa, MD, FACS, FASMBS, is Professor of Surgery at The University of California, San Francisco Fresno Medical Education Program in Fresno, California; and Past-president of the American Society for Metabolic and Bariatric Surgery.
Bariatric Times. 2018;15(9):14–20.
Introduction
Type 2 diabetes mellitus (T2DM) is a major public health problem that is significantly linked to another worldwide epidemic—obesity. Although there are many risk factors for developing T2DM, such as age, stress, certain medications, and genetics or family history, having overweight or obesity remains the single best predictor of T2DM.[1] Both T2DM and obesity are metabolic disorders characterized by defects of insulin, insulin resistance and insulin deficiency.[2] Since weight gain and body mass index (BMI) have strong relationships to diabetes and insulin resistance, it is no surprise that nearly 90 percent of people living with T2DM also have overweight or obesity.[1]
In this article, we provide statistics and predictions for both diseases, present management options for specific patient populations, describe multiple theories on why T2DM improves or, in some cases, resolves in individuals with obesity following metabolic surgery, and outline the latest consensus guidelines for T2DM treatment based on the available evidence.
The Evolution of Diabesity
The terminology surrounding obesity, T2DM, and bariatric surgery has evolved through the years. In the early days, when Drs. Edward Mason and Chikashi Ito introduced the gastric bypass, the term bariatric surgery was born—a term derived from the Greek “baros” meaning “weight.”[3,4]
Years of practice began revealing that bariatric surgery had the potential to improve metabolic syndrome, and researchers began discovering that a cluster of co-existing conditions, including high blood pressure, abdominal obesity, high triglyceride levels, and high fasting levels of blood sugar, are inextricably linked to the development of cardiovascular disease and T2DM, leading to the emergence of the term metabolic surgery. Mounting clinical evidence of the effectiveness of surgery on metabolic diseases emerged, leading the The American Society for Bariatric Surgery (ASBS) and the International Federation for the Surgery of Obesity (IFSO) to include the word “metabolic” to their names in June 2007.
Although surgical procedures are used for treatment of both obesity and diabetes, it is important to make a distinction between bariatric surgery and metabolic surgery. Bariatric surgery is defined as a procedure performed for weight control, while metabolic surgery is defined as gastrointestinal surgery performed with the goal of treating diabetes and metabolic syndrome.[5]
The concept of combining obesity and diabetes as a single entity began emerging in 1973, when Sims et al[6] reported the discovery that endocrine and metabolic changes are associated with weight gain. Over the decades, as metabolic syndrome and metabolic surgery began rising to the forefront, the words “diabetes” and “obesity” were eventually unified to create the new, more descriptive term—diabesity.
In 2017, The American Association of Clinical Endocrinologists (AACE) and American College of Endocrinology (ACE) adopted a new term—adiposity-based chronic disease (ABCD)—that explicitly identifies a chronic disease, alludes to a precise pathophysiologic basis, and avoids the stigma and confusion related to the differential use and multiple meanings of the term obesity.[7] The term ABCD was later incorporated into the IFSO mission and vision statements.
The Global Burden
Prevalence and projections. With reported cases of obesity and T2DM now at record highs, organizations around the world continue to sound the alarm about prevention and treatment in an effort to halt the growing diabesity epidemic, which time has revealed to be consistently underestimated. Since T2DM accounts for 90 to 95 percent of all diabetes cases, most estimates of diabetes as a whole are likely to be more representative of Type 2 than Type 1.[8]
In 2008, Wang et al[9] estimated that 38 percent of the world’s adult population would have overweight and 20 percent would have obesity by the year 2030. According to the most recent numbers from 2016, we’ve already reached their estimate for overweight, which is at 39 percent; the worldwide obesity rate is 13 percent.[10]
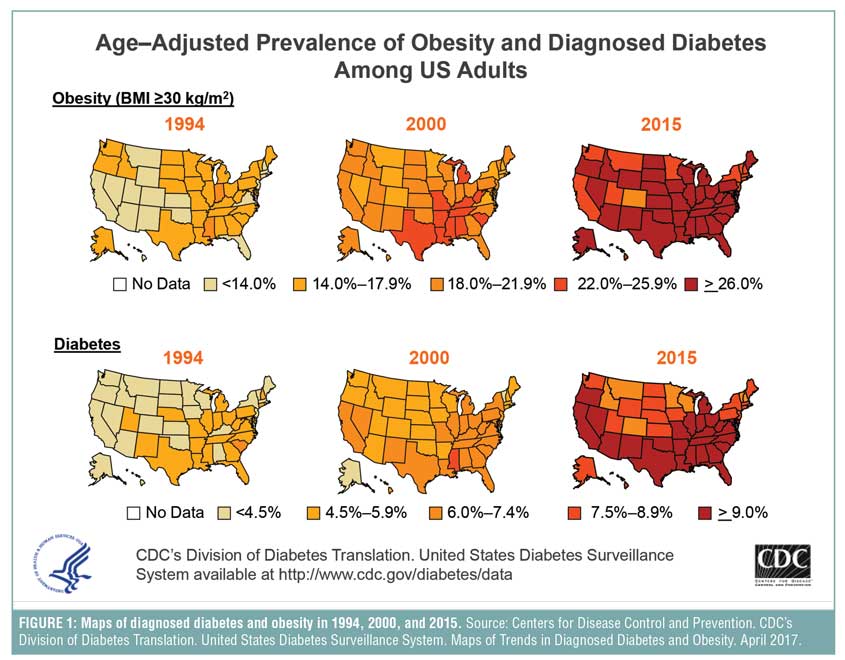
Wang et al[9] also estimated that 85 percent of the United States population would have overweight or obesity by 2030. In 2017, the Centers for Disease Control and Prevention (CDC) reported that 30.3 million Americans had diabetes (9.4%), with 23.1 million cases being diagnosed and 7.2 million cases being undiagnosed.[11] Figure 1 represents the increase of both diseases in the United States from 1994 to 2015.
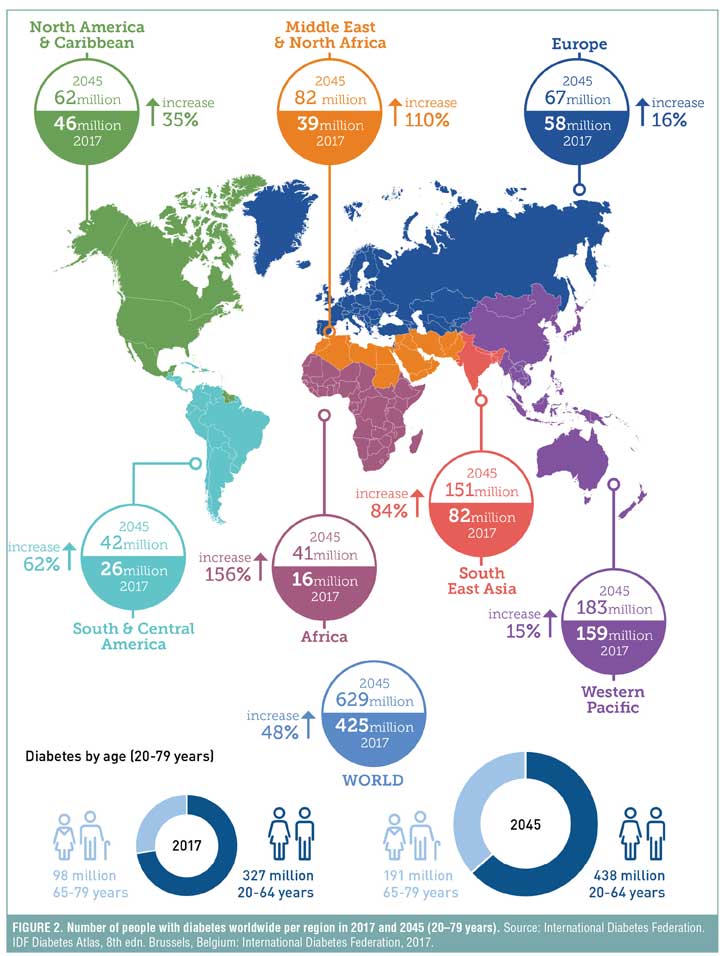
The first global predictions of a major increase in T2DM prevalence worldwide forecasted a jump between the years 1994 to 2010 from 10 million to 239 million.[12] In 2000, the International Diabetes Federation (IDF) and the World Health Organization (WHO) predicted that by 2030, 324 and 366 million people, respectively, would have diabetes worldwide.[8,13] According to the IDF’s Diabetes Atlas, a report of current and projected diabetes prevalence in 130 countries in seven regions, these predictions were gross underestimations as there were already about 415 million adults (aged 20–79 years) with diabetes in 2015 and more than 425 million in 2017. It is important to note that these numbers do not include the 212.4 million (1 in 2) individuals who are undiagnosed.[8]
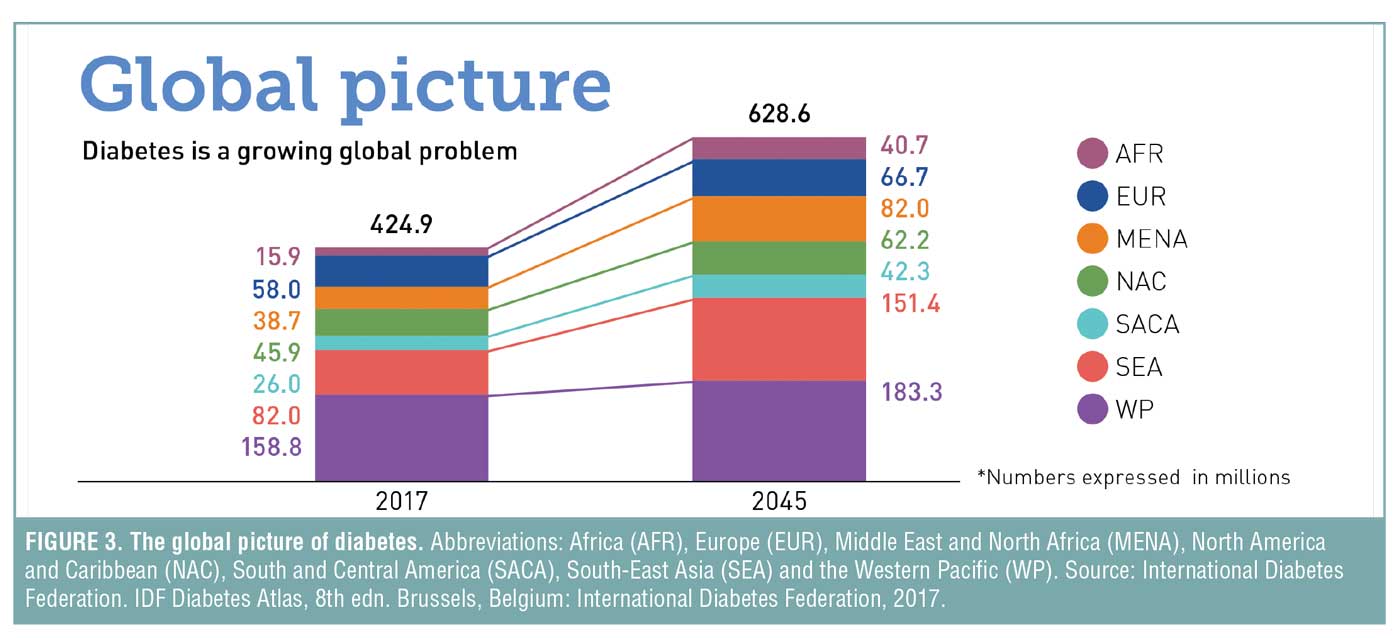
If trends continue, the IDF predicts that an estimated 629 million people worldwide will be living with diabetes by 2045,[8] and, based on history, even that might turn out to be an underestimation. This makes ABCD one of the biggest challenges of public health for the 21st century (Figures 2 and 3).
Morbidity and mortality. The risk of developing multiple comorbidities, including T2DM, significantly increases with obesity. Other risks incude cardiovascular disease, hypertension, stroke, gastroesophageal reflux disease (GERD), obstructive sleep apnea (OSA), nonalcoholic steatohepatitis (NASH), and cancer.[14] The risk of developing diabetes is almost 20 times higher in individuals who have BMIs of 35kg/m2 or higher than in those with BMIs less than 25kg/m2 (relative risk [RR]: 14–20 for women, 19–33 for men; confidence interval [CI]: 95%).[15] One study found a 20- to 30-percent increase in the risk of death among a cohort of patients with obesity, who were 50 to 71 years old at enrollment in 1995 to 1996, compared to those with normal weight.[16]
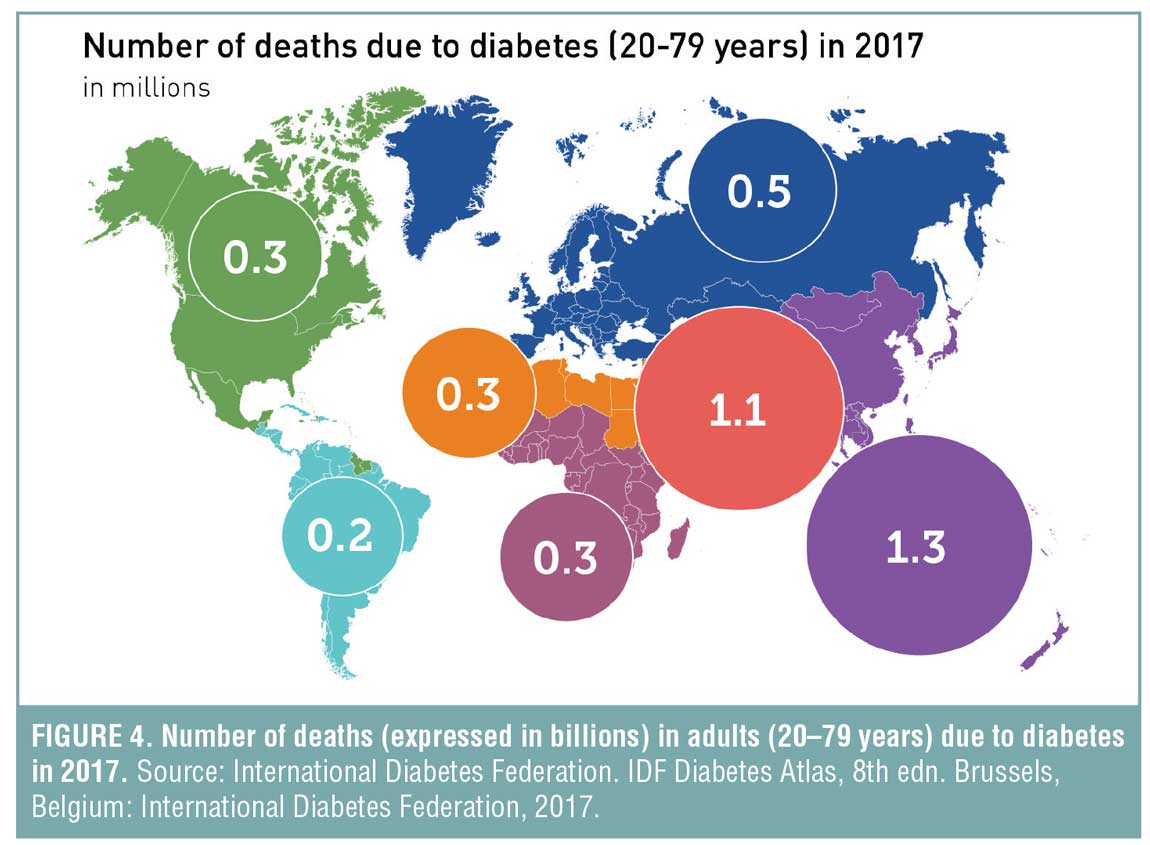
A recent study concluded that obesity and diabetes were the leading causes of preventable death in the United States, knocking tobacco use from the top spot.[17] Diabetes is currently the seventh leading cause of death, globally and nationally (Figure 4). This is significant because in 2000, diabetes was not even on the top 10 list.[18,19]
Cost of illness. The financial burden of diabesity is equally heavy considering direct (e.g., prevention, diagnosis, and treatment) and indirect costs (e.g., absenteeism).[20]
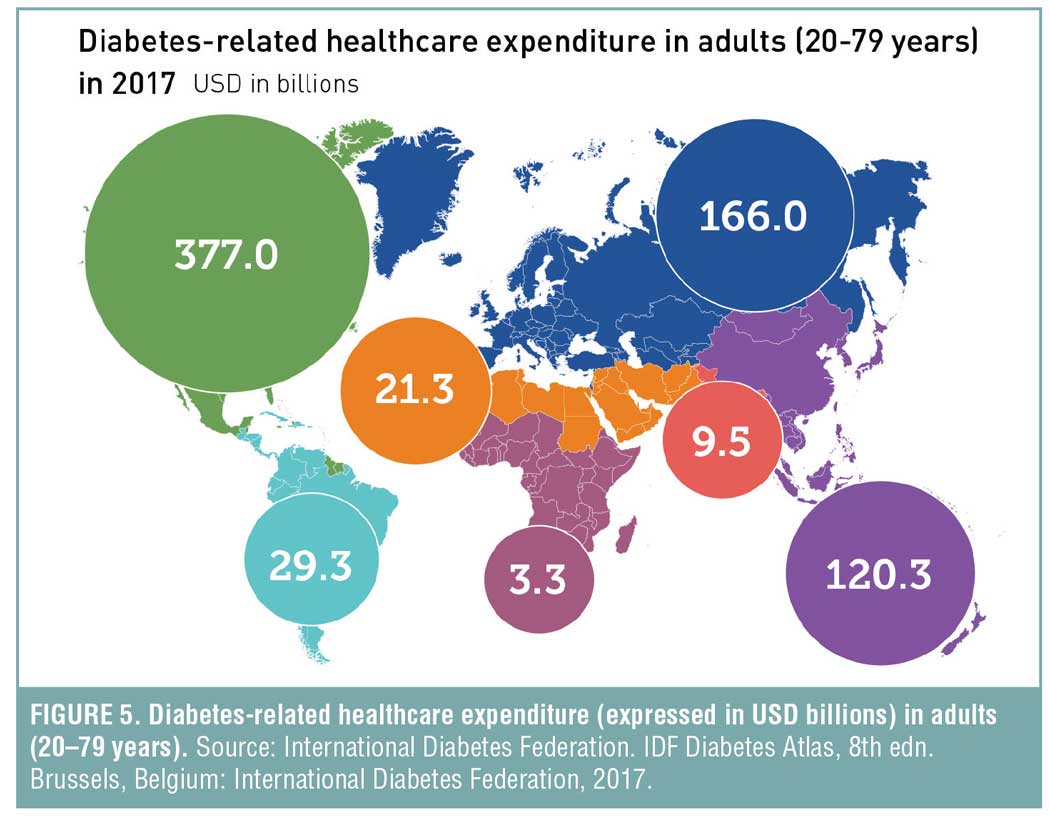
People with diagnosed diabetes, in general, have medical expenditure 2.3 times higher than those without diabetes, with expenses averaging $16,750 per year, of which $9,601 are attributed to diabetes.[21] The global economic impact of obesity was estimated to be $2.0 trillion in 2014, and expenditures for diabetes were an estimated $727 billion in 2017 (Figure 5).[8,22]
In the United States, diagnosed diabetes saw a 26-percent increase over just five years, from $245 billion in 2012 to $327 billion in 2017.[23]
Continual cost of illness (COI) assessments might help policy makers understand the economic burden of diabesity. For instance, a cost analysis of the Treat and Reduce Obesity Act (TROA) estimated that, if passed, Medicare would save between $19 billion and $21 billion over the first 10 years. This savings comes from a reduction in Part B (ambulatory care) and Part D (prescription drug) expenditures.[24]
The Diabesity Care Pathway
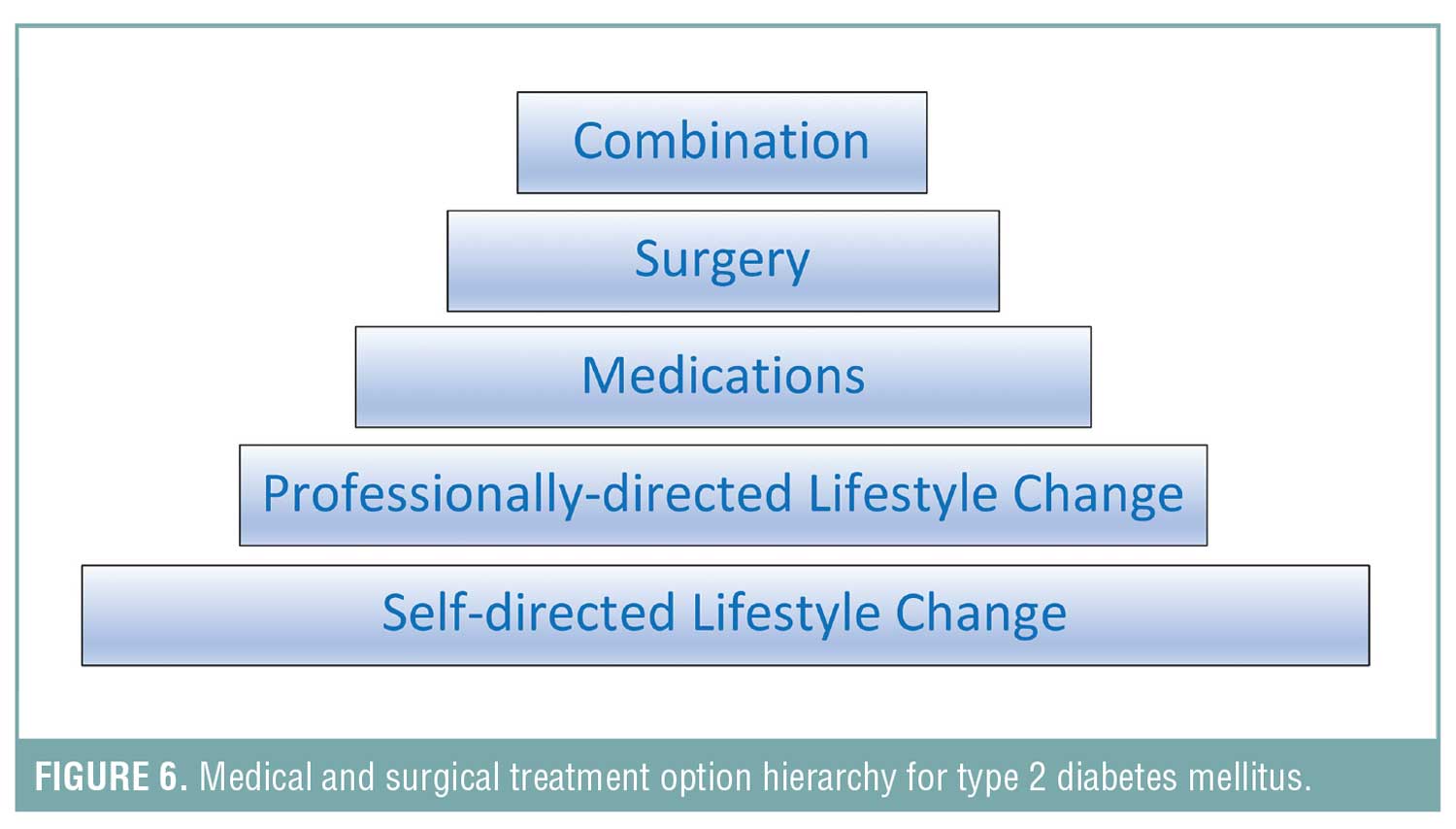
Lifestyle modifications. The management of ABCD should incorporate not only the symptoms, but also the severity and genetic characteristics of the disease. Lifestyle modifications, self or professionally directed, should provide the foundation for more invasive options and might ensure longevity for weight maintenance and cardiovascular health. Lifestyle modifications include change in diet rather than just caloric restriction, elimination or replacement of weight-promoting medications, restoration of healthy sleep cycles, and increased physical activity.[25]
In the Look AHEAD trial, individuals who lost at least 10 percent of their body weight in the first year of the study had a 21-percent lower risk of the primary outcome (adjusted hazard ratio [HR]: 0.79; 95% CI: 0·64–0·98; p=0·034) and a 24-percent reduced risk of the secondary outcome (adjusted HR 0.76; 95% CI: 0·63–0·91; p=0·003) compared with individuals with stable weight or weight gain.[26]
In people with insulin resistance, research indicates that lifestyle modifications could reduce the risk of developing diabetes up to 50 percent.[27,28]. The initial goal for patients with overweight or obesity and T2DM is to lose 5 to 10 percent of their weight within the first six months of treatment. Due to the intense need for follow-up and difficulty adhering to a new lifestyle, sustained long-term results are very limited.[5]
In our clinical experience, if lifestyle modifications fail to achieve satisfactory weight loss or weight maintenance, or if the patient has diabetes, treatment should include medications tailored to the treatment of diabetes and weight reduction.
Medication. Prospective studies comparing metformin with placebo have shown that patients taking metformin were able to lose weight for at least 10 years of treatment. This result was statistically significant compared with the placebo group.[29] Unfortunately, some common diabetic medications are associated with weight gain, especially insulin. On the other hand, the glucagon-like peptide 1 receptor (GLP-1R) agonists are associated with modest weight loss.[29]
Medications primarily targeting weight loss, such as phentermine, topirmate, lorcaserin, and combination drugs (e.g., naltrexone HCl and bupropion HCl [Contrave, Orexigen Therapeutics, Inc., La Jolla, California], phentermine and topiramate extended-release [Qsymia, Vivus, Inc, Campbell, California) can be effective for treating T2DM in some patients.30 Liraglutide [Saxenda, Novo Nordisk Inc, Plainsboro, New Jersey]), a GLP-1R agonist, was approved by the United States Food and Drug Administration (FDA) for weight loss in the absence of diabetes, but is generally cost-prohibitive for most patients at this time.
Surgery. Bariatric/metabolic surgical procedures, such as Roux-en-Y gastric bypass (RYGB), have been shown to be effective not only with absolute weight loss, but also with weight maintenance. Compared with medical management alone, these surgical procedures have direct effects on glucose metabolism and reduce the micro- and macrovascular complications associated with diabetes.[31] In addition, bariatric surgery has been shown to significantly improve survival in patients with obesity compared with those who didn’t undergo surgery.[32,33]
The efficacy of bariatric surgery for weight loss and T2DM remission in descending order is as follows: biliopancreatic diversion with or without duodenal switch (BPD+/-DS), RYGB, sleeve gastrectomy (SG), and laparoscopic adjustable gastric banding (LAGB).[34]
Combination therapy. Complete resolution of diabetes may not always be possible. There are many factors that can influence the chances of remission, such as the surgical procedure, duration of diabetes, preoperative glycemic control, and number of medications taken preoperatively. For this reason it is not uncommon that a combination of surgical and medical therapy might be used.[35]
Since metabolic changes occur immediately after bariatric surgery independently of weight loss, it is important to titrate down a patient’s diabetes medications postoperatively because there is a higher propensity to hypoglycemia during this period. For patients who were on insulin preoperatively, the short-acting insulin should be decreased by at least 50 percent, and long-acting insulin can be stopped if the dose was less than 30 units or decrease by more than 50 percent if it was higher 30 units.[36]
Metformin can be continued as a stand-alone therapy in the postoperative period for patients with HbA1c less than nine percent. If HbA1c is higher than nine percent, a second oral agent can be administered. Long-acting sulfonylureas and thiazolidinediones are discouraged in general due to the risk of hypoglycemia or decreasing the bone density, respectively. Long-term management with sodium-glucose cotransporter-2 (SGL2) inhibitors and e glucagon-like peptide-1 (GLP-1) analogues have been favored as well as an adjusted insulin regimen.[36]
These are all general recommendations, however, individualized treatment should be tailored based on a patient’s response and glycemic levels.
Metabolic Surgery: Mechanisms of Action: Proposed Theories
There are multiple theories on why diabetes improves or, in some case, resolves, in individuals with obesity following metabolic surgery. These theories are not necessarily related to weight loss since many of the changes in glucose control are seen before there is significant weight loss. The mechanisms of action are still not completely understood, and no a single theory is considered an absolute truth. The reality is that bariatric surgery causes a complex interaction of many known and certainly some unknown metabolic factors.
In order to understand some of the mechanistic processes of the effect of bariatric surgery on T2DM, it is important to define incretins. Incritins are intestinal hormones that promote postprandial secretion of insulin. Two major peptides have been identified in the mechanistic processes of bariatric surgery—gastric inhibitory peptide (GIP), which are secreted by the K cells in the duodenum, and GLP-1, which is secreted by the L cells in the ileum. GLP-1 stimulates insulin secretion, proliferation of pancreatic beta cells, and increases gluconeogenesis in the liver and skeletal muscle. GIP stimulates beta cells and inhibits their apoptosis. GIP also stimulates lipoprotein lipase activity.
Metabolic theories. There are two main theories relating to the metabolic effects of various bariatric surgical procedures. Other incretins and naturally occurring substances and microorganisms likely influence these two major theories: the foregut theory and hindgut theory.
The hypothesis of the foregut theory is that there are “anti” incretins in the proximal small bowel that lead to abnormal glycemic control, theoretically to prevent hypoglycemia. With surgeries like the RYGB or BPD, this region is bypassed and the secretion from those substances avoided.[37]
The hypothesis of the hindgut theory is that the rapid delivery of nutrients to the ilium stimulates secretion of GLP-1 and thus helps to better regulate blood sugar and improve diabetic control.[37]
Ghrelin. Ghrelin is an incretin that is also known as the “hunger hormone” and is secreted mainly at the gastric fundus. It is believed to not only increase hunger, but to also inhibit insulin secretion. Most cells producing ghrelin are removed in the SG procedure, and the fundus is bypassed in RYGB.
Bile acids. Bile acids play a role in the digestion and absorption of fat and liposoluble vitamins and are involved in the enterohepatic circulation (EHC), which is the movement of bile acid molecules from the liver to the small intestine and back to the liver. Bile acids interact with intranuclear receptors in the liver, inhibiting gluconeogenesis and promoting release of GLP-1. Some studies have shown that levels of bile acids increase after SG.[38]
Gut flora. Human gut flora is dominated by two major groups of bacteria: Bacteroides and Firmicutes. Research has shown that there is a larger presence of Firmicutes in patients with obesity. Studies in mice models have demonstrated that insulin sensitivity improved after transferring intestinal flora from lean mice donors to insulin-resistant mice. Certain microbiota also induce the breakdown of substances like fatty acids, which could lead to an increase of GLP-1 and peptide YY (PYY). PYY stimulates the vagus nerve to produce satiety.[39]
Evidence, Consensus Statement, and Guidelines
Randomized controlled trials and Diabetes Surgery Summit II (DSS-II). The Diabetes Surgery Summit II (DSS-II) in 2015 was an international consensus meeting of 48 experts comprising diabetologists, endocrinologists, internal medicine physicians, cardiologists, gastroenterologists, primary care physicians, dietitians, and surgeons, and included official representatives of partner diabetes organizations. This panel established guidelines that are endorsed by more than 46 different professional societies, including IFSO, American Association of Clinical Endocrinologists (AACE), American College of Surgeons (ACS), American Society for Metabolic and Bariatric Surgery (ASMBS), and The Obesity Society (TOS).[40] Panel members addressed a wide range of issues, including mechanisms of action by which surgery improves diabetes, and the effects that glucose control and surgical intervention have on micro- and macrovascular complications of the human metabolism.
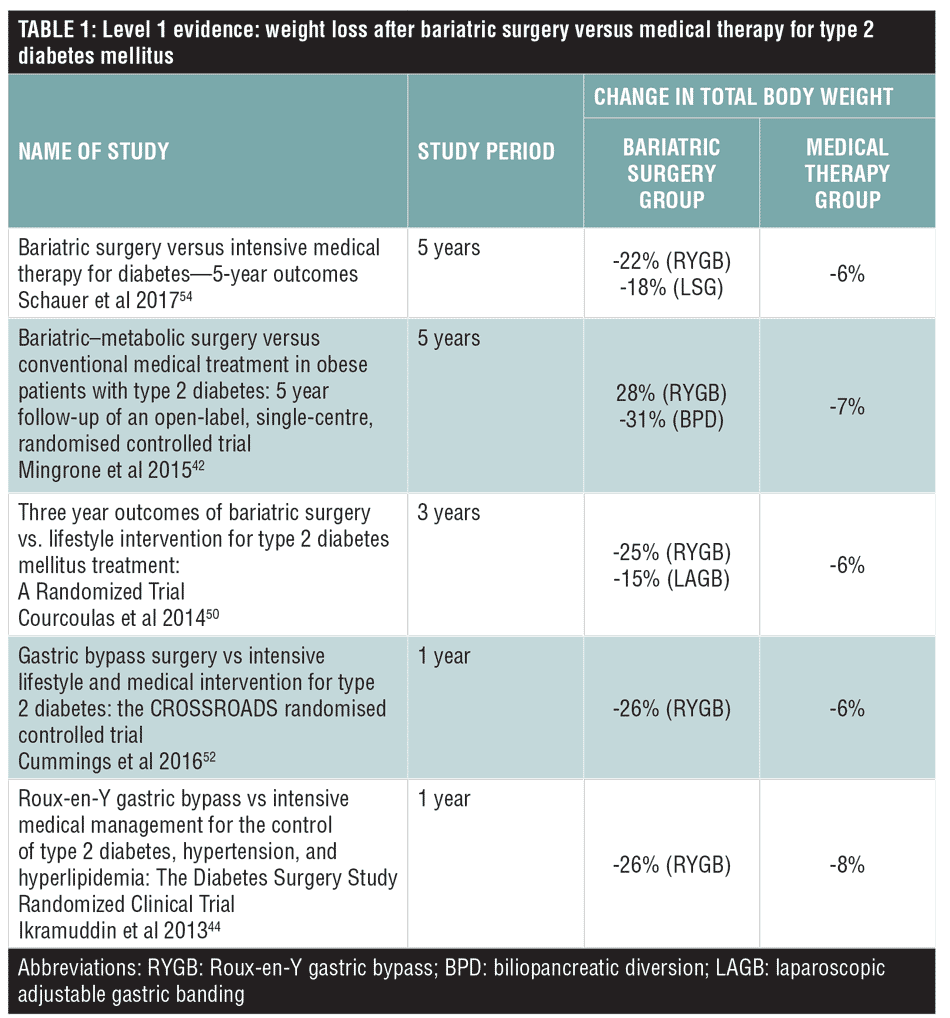
At the time when the DSS-II guidelines were developed, there were 11 randomized controlled trials (RCTs) comparing metabolic surgery with lifestyle modification and medication management.[41–52] A total of 794 patients were included in these studies. Surgical treatment was found to be more effective for remission of diabetes and weight loss compared with nonsurgical interventions, and in all but one of the studies,51 results were statistically significant in favor of bariatric surgery.
In a study by Ding et al[51] remission of T2DM was achieved in 33 percent for the surgery group compared with 23 percent in the medical weight loss alone group. The effectiveness of bariatric surgery in this study was lower than what is typically seen, likely because LAGB was the surgical procedure used, and this the least effective of the metabolic procedures.[53]
Bariatric surgery has been shown to reduce hemoglobin A1C (HbA1C). Most of the 11 studies that were available at the time of the DSS-II demonstrated superiority in the surgery groups compared to the nonsurgical groups in secondary outcomes such as increased high-density lipoprotein (HDL), lower triglycerides, and improvement in quality of life (QoL).
In the Surgical Treatment and Medications Potentially Eradicate Diabetes Efficiently (STAMPEDE) trial, Schauer et al[54] compared intensive medical treatment to SG and RYGB procedures among 150 patients. Five-year results of this randomized controlled study showed that surgery was significantly more effective than intensive medical therapy for resolution of diabetes, defined as HbA1C level of 6.0% or less, with or without the use of diabetes medications. Surgery was also superior in terms of weight reduction, medication reduction, improvement in lipid levels, and quality of life. The resolution rate of T2DM was 29 percent and 23 percent in the RYGB and SG groups, respectively, compared with five percent in the medical therapy group (P<0.03). Results also demonstrated that the RYGB was associated with greater weight loss and reduction in use of diabetes medications compared to weight loss alone; however, the trial was not powered sufficiently to detect significant differences between the two procedures, and the authors concluded that larger trials with longer follow-up are required.[54]
Since the DSS-II Consensus guidelines were released, Schauer et al[54] released the five-year data from the STAMPEDE study. Tables 1 and 2 list the latest Level 1 evidence for the role of bariatric surgery compared with intensive medical therapy alone for weight loss and T2DM management.[55]
Main recommendations from the DSS-II Consensus include the following:
There is sufficient clinical evidence that supports the inclusion of metabolic surgery in the management of diabesity and treatment algorithms that include surgery should be considered (Figure 6).
- RYGB, SG, LAGB, and BPD +/-DS each have their own risk/benefit ratio. Any other procedure is still considered investigational. Recommendations on patient selection for metabolic surgery include the following:
-Class III obesity (BMI ≥40kg/m2)
-Class II obesity(BMI 35.0–39.9kg/m2) with inadequate glycemic control
-Class I obesity (BMI 30.0–34.9kg/m2) with inadequate glycemic control despite optimal medical treatment by either oral or injectable medication. - RYGB appears to have a more favorable risk/benefit profile in most patients with T2DM.
- SG is an effective procedure for weight loss and control of T2DM, at least in the short and medium term. Long-term studies are required.
- LAGB is effective in improving glycemic control to the degree that it causes weight loss; however, this procedure has no metabolic effect when used alone and is associated with a greater risk of revisions and complications.
- BPD +/- DS is the most effective procedure in terms of glycemic control and weight loss; however, it’s associated with a greater risk of nutritional deficiencies, which make it less favorable than other metabolic procedures and should be considered only in patients with super obesity (BMI≥50kg/m2).
- After surgery, a multidisciplinary team with expertise in diabetes should manage the patients.[40]
Barriers
It has been estimated that about 80 percent of people with diabetes live in low- or middle-income communities.[8] Access to metabolic surgery in these populations might be limited, with cost being the primary barrier. Despite the proven track record of bariatric/metabolic surgery as it pertains to safety, efficacy and durability; a low number of patients are referred to bariatric/metabolic surgeons.[56] This could be due to misconceptions about the risk of surgery versus the natural progression of the disease itself, inadequate knowledge about diabetes remission with metabolic surgery, and simple dismissal of the well-documented outcomes with metabolic/bariatric surgery.[56]
The ADA and the IDF helped develop and have ratified the DSS-II consensus statements and guidelines, and several other organizations have formally endorsed the DSS-II consensus statements and guidelines. This is significant when one considers that just a few years ago, metabolic surgery was not in the treatment algorithms of the major diabetes treatment organizations. It was not until 2009 that the ADA included metabolic surgery in their annual guidelines, but even then, the recommendations were based on weight, not T2DM management.
They also included lower weight-based parameters in their recommendations, in order to address the Asian populations who have a higher risk of diabetes for the same basal metabolic index.[57]
Participants at the Policy Lab, held during the 3rd World Congress on Interventional Therapies For Type 2 Diabetes, identified four building blocks to facilitate change in practice and policy, with the ultimate goal of increasing the use of metabolic surgery for the patients who can benefit.[56]. The building blocks are: 1) communicate scale of diabetes challenge; 2) properly articulate the role for bariatric/metabolic surgery; 3) identify cost effectiveness/savings; 4) explore resources/processes to support surgery access.
Conclusion
The belief that diabesity is avoidable through self-control and caloric restriction needs to be dismantled. This can be achieved through constant education. With continued lobbying and policy change, access to care, and referrals for metabolic/bariatric surgery by other physician groups will improve, ultimately helping clinicians achieve optimal results among the diabesity population.
References
- World Health Organization. Obesity and Overweight Fact Sheet. http://www.who.int/dietphysicalactivity/media/en/gsfs_obesity.pdf Accessed August 30, 2018.
- Verma S, Hussain ME. Obesity and diabetes: An update. Diabetes Metab Syndr. 2017;11(1):73–79. Epub 2016 Jun 17.
- Mason, EE, Ito C. Gastric bypass in obesity. Surg Clin North Am. 1967;47:1345–1351.
- Deitel M, Melissas J. The origin of the word “bari”. Obes Surg. 2005;15(7):1005–1008.
- Gadde KM, Martin CK, et al. Obesity: pathophysiology and management. J Am Coll Cardiol. 2018;71(1):69–84.
- Sims EA, Danforth E Jr, Horton ES, Bray GA, Glennon JA, Salans LB. Endocrine and metabolic effects of experimental obesity in man. Recent Prog Horm Res. 1973;29:457–496.
- Mechanick JI, Hurley D, Garvey WT. Adiposity-based chronic disease as a new diagnostic term: The American Association of Clinical Endocrinologists and American College of Endocrinology position statement. Endocr Pract. 2017;23(3):372–378.
- International Diabetes Federation. IDF Diabetes Atlas, 8th edn. Brussels, Belgium: International Diabetes Federation, 2017
- Wang Y, Beydoun MA, et al. Will all Americans become overweight or obese? Estimating the progression and cost of the US obesity epidemic. Obesity (Silver Spring). 2008; 16(10):2323–2330.
- World Health Organization. Obesity and overweight: key facts. February 16, 2018. http://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight. Accessed August 27, 2018.
- Centers for Disease Control and Prevention. National Diabetes Statistics Report, 2017. Atlanta, GA: Centers for Disease Control and Prevention, US Department of Health and Human Services; 2017.
- Zimmet P, McCarty D. The NIDDM epidemic: global estimates and projections—a look into the crystal ball. IDF Bulletin. 1995;40:8–16.
- Zimmet PZ. Diabetes and its drivers: the largest epidemic in human history? Clin Diabetes Endocrinol. 2017;3:1.
- Schauer D, et al. Bariatric surgery and the risk of cancer in a large multisite cohort. Ann Surg. 2017 Sep 21.[Epub ahead of print.
- Field AE, Coakley EH, Must A, et al. Impact of overweight on the risk of developing common chronic diseases during a 10-year period. Arch Intern Med. 2001;161:1581–1586.
- Adams KF, Schatzkin A, Harris TB, et al. Overweight, obesity, and mortality in a large prospective cohort of persons 50 to 71 years old, N Engl J Med. 2006;355:763–778.
- Cleveland Clinic. Obesity is top cause of preventable life-years lost, study shows. ScienceDaily. April 22, 2017. www.sciencedaily.com/releases/2017/04/170422101614.htm. Accessed August 30, 2018.
- Global Health Estimates 2016: Disease burden by Cause, Age, Sex, by Country and by Region, 2000-2016. Geneva, World Health Organization; 2018.
- Murphy SL, Xu J, Kochanek KD, Curtin SC, Arias E. Deaths: final data for 2015. Natl Vital Stat Rep. 2017;66(6):1–75.
- Hammond RA, Levine R. The economic impact of obesity in the United States. Diabetes Metab Syndr Obes. 2010;3:285–295.
- American Diabetes Association. Economic Costs of Diabetes in the U.S. in 2017. Diabetes Care. 2018;41(5):917–928.
- Tremmel M, Gerdtham UG, Nilsson PM, Saha S. Economic burden of obesity: a systematic literature review. Int J Environ Res Public Health. 2017;14(4):pii: E435.
- American Diabetes Association. Economic Costs of Diabetes in the U.S. in 2017. Diabetes Care. 2018;41(5):917–928.
- Su Wayne. The budgetary impact of expanding Medicare’s coverage of anti-obesity interventions. IHS Markit. Accessed August 30, 2018.
- Wadden TA, Webb VL, Moran CH, Bailer BA. Lifestyle modification for obesity: new developments in diet, physical activity, and behavior therapy. Circulation. 2012;125(9):1157–1170.
- Look AHEAD Research Group. Association of the magnitude of weight loss and changes in physical fitness with long-term cardiovascular disease outcomes in overweight or obese people with type 2 diabetes: a post-hoc analysis of the Look AHEAD randomised clinical trial. Lancet Diabetes Endocrinol. 2016;4(11):913–921.
- Knowler WC, Barrett-Connor E, Fowler SE, et al. Diabetes Prevention Program Research Group.: reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med. 2002;346(6):393e403.
- Tuomilehto J, Lindstrom J, Eriksson JG, et al. Finnish Diabetes Prevention Study Group: prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N Engl J Med. 2001;344(18):1343e50.
- Diabetes Prevention Program Research Group. Long-term safety, tolerability, and weight loss associated with metformin in the Diabetes Prevention Program Outcomes Study. Diabetes Care. 2012;35:731–737.
- MacDaniels JS, Schwartz TL. Effectiveness, tolerability and practical application of the newer generation anti-obesity medications. Drugs Context. 2016;5:212291.
- Adams TD, Arterburn DE, Nathan DM, Eckel RH. Clinical outcomes of metabolic surgery: microvascular and macrovascular complications. Diabetes Care. 2016;39(6):912–923.
- Adams T, Gress RE, Smith SC, et al. Long-term mortality after gastric bypass surgery. N Engl J Med. 2007; 357:753–761.
- Sjöström L. Review of the key results from the Swedish Obese Subjects (SOS) trial—a prospective controlled intervention study of bariatric surgery. J Intern Med. 2013;273(3):219–234. Epub 2013 Feb 8.
- Koliaki C, Liatis S, le Roux CW, Kokkinos A. The role of bariatric surgery to treat diabetes: current challenges and perspectives. BMC Endocr Disord. 2017;17(1):50.
- Dixon JB, Zimmet P, Alberti KG, Rubino F, International Diabetes Federation Taskforce on Epidemiology and Prevention. Bariatric surgery: an IDF statement for obese type 2 diabetes. Diabet Med. 2011;28(6):628–642.
- Kheniser KG, Kashyap SR. Diabetes management before, during, and after bariatric and metabolic surgery. J Diabetes Complications. 2018;32(9):870–875.
- Mingrone G, Castagneto-Gissey L. Mechanisms of early improvement/resolution of type 2 diabetes after bariatric surgery. Diabetes Metab. 2009;35(6 Pt 2):518–523.
- Tomkin GH, Owens D. Obesity diabetes and the role of bile acids in metabolism. J Transl Int Med. 2016;4(2):73–80. Epub 2016 Jul 7
- Khoury LE, Chouillard E, Chahine E, et al. Metabolic surgery and diabesity: a systematic review. Obes Surg. 2018;28(7):2069–2077.
- Rubino F, Nathan DM, Eckel RH, et al. 2nd Diabetes Surgery Summit. Metabolic surgery in the treatment algorithm for type 2 diabetes: a joint statement by international diabetes organizations. Diabetes Care. 2016;39:861–877.
- Dixon JB, O’Brien PE, Playfair J, Chapman L, Schachter LM, Skinner S, et al. Adjustable gastric banding and conventional therapy for type 2 diabetes: a randomized controlled trial. JAMA. 2008;299:316–323.
- Mingrone G, Panunzi S, De Gaetano A, et al. Bariatric surgery versus conventional medical therapy for type 2 diabetes. N Engl J Med. 2012;366:1577–1585.
- Schauer PR, Kashyap SR, Wolski K, et al. Bariatric surgery versus intensive medical therapy in obese patients with diabetes. N Engl J Med. 2012;366:1567–1576.
- Ikramuddin S, Korner J, Lee WJ, et al. Roux-en-Y gastric bypass vs intensive medical management for the control of type 2 diabetes, hypertension, and hyperlipidemia: the diabetes surgery study randomized clinical trial. JAMA. 2013;309:2240–2249.
- Abdeen G, le Roux CW. Mechanism underlying the weight loss and complications of Roux-en-Y gastric bypass. Review Obes Surg. 2016;26:410–421.
- Halperin F, Ding SA, Simonson DC, et al. Roux-en-Y gastric bypass surgery or lifestyle with intensive medical management in patients with type 2 diabetes: feasibility and 1-year results of a randomized clinical trial. JAMA Surg. 2014;149:716–726.
- Parikh M, Chung M, Sheth S, et al. Randomized pilot trial of bariatric surgery versus intensive medical weight management on diabetes remission in type 2 diabetic patients who do NOT meet NIH criteria for surgery and the role of soluble RAGE as a novel biomarker of success. Ann Surg. 2014;260:617–622. discussion 622–624.
- Schauer PR, Bhatt DL, Kirwan JP, et al.; STAMPEDE Investigators. Bariatric surgery versus intensive medical therapy for diabetes—3-year outcomes. N Engl J Med. 2014;370:2002–2013.
- Wentworth JM, Playfair J, Laurie C, et al. Multidisciplinary diabetes care with and without bariatric surgery in overweight people: a randomised controlled trial. Lancet Diabetes Endocrinol. 2014;2:545–552.
- Courcoulas AP, Belle SH, Neiberg RH, et al. Three-year outcomes of bariatric surgery vs lifestyle intervention for type 2 diabetes mellitus treatment: a randomized clinical trial. JAMA Surg. 2015;150:931–940.
- Ding SA, Simonson DC, Wewalka M, et al. Adjustable gastric band surgery or medical management in patients with type 2 diabetes: a randomized clinical trial. J Clin Endocrinol Metab. 2015;100:2546–2556.
- Cummings DE, Arterburn DE, Westbrook EO, et al. Gastric bypass surgery vs intensive lifestyle and medical intervention for type 2 diabetes: the CROSSROADS randomised controlled trial. Diabetologia. 2016;59:945–953.
- Schauer PR, Mingrone G, Ikramuddin S, Wolfe B. Clinical outcomes of metabolic surgery: efficacy of glycemic control, weight loss, and remission of diabetes. Diabetes Care. 2016; 39(6):902–911.
- Schauer PR, Bhatt DL, Kirwan JP, et al. Bariatric surgery versus intensive medical therapy for diabetes—5-year outcomes. N Engl J Med. 2017;376(7):641–651.
- Nguyen N, Varela E. The dynamic and evolving field of metabolic and bariatric surgery: current perspective. Bariatric Times. 2017;14(4):16–17.
- Rubin JK, Hinrichs-Krapels S, Hesketh R, et al. Idenfifying barriers to appropriate use of metabolic/bariatric surgery for type 2 diabetes treatment: Policy Lab results. Diabetes Care. 2016; 29:954–963.
- Rhee EJ. Diabetes in Asians. Endocrinol Metab (Seoul). 2015;30(3):263–269.
Category: Past Articles, Review