Laparoscopic Gastric Bypass Conversion to Duodenal Switch
 by Peter C. Ng, MD; Lindsey S. Sharp, MD; and Dustin M. Bermudez, MD
by Peter C. Ng, MD; Lindsey S. Sharp, MD; and Dustin M. Bermudez, MD
Drs. Ng, Sharp, and Bermudez are with Rex Bariatric Specialists, UNC/Rex Healthcare in Raleigh, North Carolina.
FUNDING: No funding was provided.
DISCLOSURES: The authors have no conflicts of interest relevant to the content of this article.
ABSTRACT: We present a review of laparoscopic conversion of gastric bypass to duodenal switch (DS) as a treatment alternative for disease recurrence after gastric bypass. We discuss the technical aspects of conversions, the limitations to adoption, the current published experience and outcomes, recommendations for patient selection, potential technical challenges and complications, and the nutritional management after DS. While DS offers a viable alternative as a strategy to manage gastric bypass failure with acceptable weight loss, careful technical planning, patient selection, and long-term nutritional care remain critical to prevent postoperative surgical and long-term nutritional problems.
KEYWORDS: Bariatric surgery, revision, gastric bypass, duodenal switch, malabsorption
Bariatric Times. 2020;17(9):16–17.
Duodenal switch is an effective surgical strategy for inadequate weight loss and comorbidity recurrence after gastric bypass. Longitudinal studies of gastric bypass report rates of disease recidivism as high as 35 percent.1 Considering the estimated 362,000 gastric bypasses performed between 2011 and 2018, the potential need for revision options count in the thousands.2
Conventional surgical approaches to revision after gastric bypass typically focus on gastric restriction. These techniques produce inconsistent and generally modest results. In comparison, conversion from gastric bypass to a malabsorptive procedure shows promise, demonstrating superior weight loss and effective comorbidity resolution. Access to laparoscopic conversion of gastric bypass to duodenal switch (DS) is limited, however, by lack of surgeon awareness, experience, or acceptance, technical complexity, and apprehension over nutritional consequences and patient compliance.
In this review, we discuss in detail the fundamental concepts around laparoscopic bypass conversion to DS (GBDS), including a critical examination of the current revision options after bypass, GBDS surgical technique, weight, and comorbidity outcomes in the literature, as well as complications and nutritional results.
Bypass to DS Technique
The GBDS conversion consists of three defined stages with each representing a potential stopping point: 1) reconstruction of the gastric reservoir, 2) duodenal division and duodenoileostomy, and 3) ileoileostomy/reversal of the roux limb. While most reoperative surgery is inherently more complicated, GBDS introduces a higher risk profile. Multiple anastomoses, a long and tedious operation, and inexperience with gastrogastrostomy and DS techniques limit wider adoption among bariatric surgeons. Thoughtful consideration and surgical planning is critical to safe adoption.
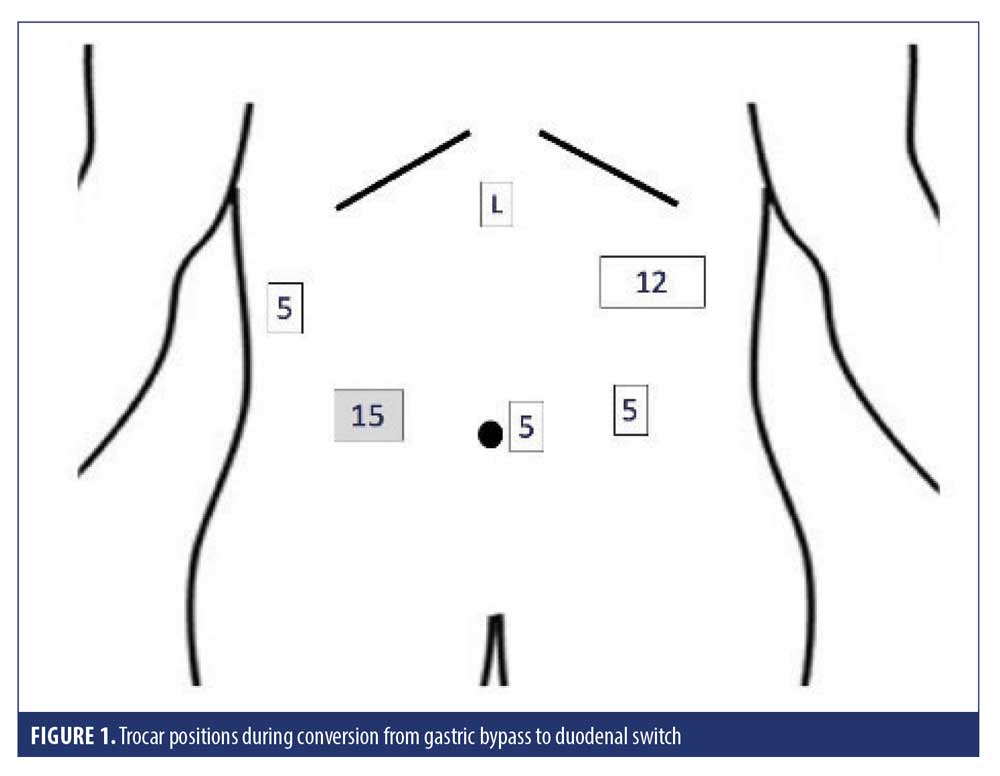
Once trocars are placed (see Figure 1), the roux limb is identified, mobilized, and measured. The roux limb is stapled and divided approximately 5cm distal to the gastrojejunal anastomosis with a 60mm stapler, and the blood supply transected with bipolar energy, allowing the small bowel aspect of the gastrojejunal stoma to demarcate. The remnant and gastric pouch are dissected free and separated, preserving the left gastric and lesser curve vasculature. The pouch is divided with (black or green load) endostapler just above the stoma, removing the stoma and detached proximal roux limb. Preoperative endoscopy is valuable to assure adequate pouch size for reconstruction and plan gastric reconstruction, particularly if the pouch appears small potentially limiting reach and compromising stapling.
The gastrogastrostomy is considered the most technically challenging element of GBDS conversion. The short gastric vessels along the fundus are divided, preserving the distal 10cm of antral greater curve mesentery. Preserving this antral blood supply allows the reconstructed stomach to remain well vascularized, minimizing risk of ischemia and leak. The remnant stomach is then stapled transversely at the incisura to remove the fundus. We reconstruct the gastrogastrostomy using a linear stapled technique, aligning the antrum and pouch longitudinally with a 60mm stapler (purple or yellow) advanced to at least 40mm. We pass a 40 F bougie to assure patency and close the common gastrogastrostomy using a single layered hand sewn technique with absorbable suture.
Next, the distal small bowel is measured and marked retrograde from the terminal ileum for 300cm, oriented with the biliopancreatic limb lying medially. After assuring the appropriate reach for this omega loop, we affix the loop to the omentum temporarily. We proceed to dividing the duodenum just distal to the duodenal bulb. A retroduodenal dissection technique is important to intentionally maintain the distal antral blood supply. The retroduodenal tunnel is formed 3 to 4cm distal to the pylorus (typically at the level of the common bile duct) with a 60mm load stapler (tan or white).
The duodenoileostomy is constructed, in our practice, using a fully stapled technique.3 While the hand-sewn technique is more broadly practiced, it requires a substantial learning curve and longer operative times. We found greater consistency and efficiency with our stapled technique, noting exceedingly low risk of leak and no strictures in our larger experience.4 We use a 45 staple load (tan or white) to create an end-to-side anastomosis to the duodenal bulb, carefully orienting the intestinal loop with the biliopancreatic limb lying medially. We typically achieve a 30 to 35mm lumen. The remaining enteroenterotomy is closed with a 60mm tan load stapler. If the first staple fire reaches less than 25mm, we hand sew the common enterotomy closed. In either technique, an apical corner security suture is placed using an absorbable suture. We test each anastomosis with a provocative air leak test to 45mmHg.
If advancing to a formal DS as opposed to a single anastomosis duodenoileostomy (SADI), we divide the biliopancreatic limb with a 45mm tan load stapler. The small bowel mesentery is preserved with little to no division. The roux limb is measured 200cm distal to the duodenoileostomy and an Ileoileostomy formed with a bidirectional 45mm tan load technique and closed longitudinally with a 60mm tan load, to form a 100cm distal common channel.
Finally, we measure the gastric bypass roux limb. If short (<50 cm), we leave this in place. If longer, we will re-anastomose the roux to the biliopancreatic limb to re-establish continuity of the proximal small bowel.
All specimen are removed using a protective bag to avoid contamination. The extraction 15mm trocar site wound is washed with betadine.
We have recognized a pattern of early postoperative nausea related to dysfunctional gastric emptying and reflux. We theorize that the vagal nerve palsy from gastric bypass, hiatal or lesser curve dissection, and/or remnant exclusion lead to indolent pyloric function. Prior to closing, we selectively perform a botulinum toxin injection (100 units) at the pylorus. We decide in favor of injection if we see delayed relaxation of the pylorus during leak testing, typically characterized as delayed insufflation past the pylorus into the bulb and duodenoileostomy. If recognized postoperatively, we perform endoscopy for pyloric dilation and botulinum toxin injection as an outpatient.
Published Outcomes
In the literature, several early series first described the feasibility of GBDS, and demonstrate similar early weight loss outcomes. Keshishian et al5 published the first series conversions to DS with 46 patients (15 vertical banded gastroplasty to DS, 31 GBDS) with 67 percent excess weight loss (EWL) at 30 months. Parikh et al6 published their early experience with 12 conversions, noting an average EWL% of 63 percent at 11 months with no mortalities or leaks described. One patient required laparotomy for adhesions, and four patients developed stricture at the gastrogastrostomy (circular technique).
More recent reports demonstrate similar outcomes. Surve et al7 reported their experience of 9 patients with GBDS conversion. EWL reached 68 percent at two years, and 4 of 9 patients experienced early complications: one abdominal abscess, one leak at the gastrogastrostomy, one leak at a small bowel enterotomy, and one acute blood loss anemia. Halawani et al8 also reported a series of nine patients, noting similar results with 64 percent EWL at a 16-month follow-up. They described no short-term complications, reoperation or readmission over 30 days postoperatively, and no leaks or mortality. Moon et al9 reported 15 patients who were converted from gastric bypass to SADI-S (10 patients) and GBDS (5 patients) with a 13 percent leak rate (duodenal stump in SADI-S and sleeve leak in biliopancreatic diversion with DS [BPD-DS]). Overall EWL for the cohort was 58 percent at a two-year follow-up.9 Patients who underwent conversion to BPD-DS had greater reduction in body mass index (BMI) compared to patients who underwent SADI-S and an overall greater rate of weight loss. For the cohort, diabetes mellitus (DM) remission rate was 100 percent (7 of 7 patients), and hypertension (HTN) resolved in 40 percent (2 of 5 patients).
Complications
Complications related to DS, and specifically GBDS conversion, remain an important consideration, and awareness is critical to preventing perioperative morbidity. Perioperative complications, such as bleeding, wound infection, leak, venous thrombosis, and pulmonary embolus, occur rarely. The most common early issue is postoperative nausea and vomiting, leading to potential dehydration and moderate protein calorie deficiency. As with gastric bypass and sleeve gastrectomy, refractory nausea might lead to poor oral intake. However, the added distal bypass limits the ability for patients to self-recover without support. Aggressive outpatient rehydration, weekly or more frequent follow-up, dietician support, and surgeon assessment remain the mainstay of early identification and recovery.
As previously noted, we elect to selectively treat the pylorus with botulinum toxin to prevent early postoperative nausea and vomiting. This grew from our experience, noting a pattern of delayed gastric emptying to solids and postoperative nausea after conversion. Based on Cerfolio et al,10 we endoscopically inject of 100 units into the pylorus to relax the muscle.
Late complications most typically pertain to protein calorie malnutrition, fat soluble vitamin deficiencies, and hypokalemia. Typically, hypoalbuminemia manifests as fatigue, lower extremity edema, and muscle mass loss. Occurring in less than two percent of patients, this complication requires aggressive nutritional counseling and serial objective testing. We approach this first with oral protein supplementation and pancreatic enzyme replacement therapy and, if unsuccessful, we proceed to nutritional repletion with enteral or parenteral nutrition. Ultimately, patients failing to progress off enteral or parenteral nutrition are candidates for lengthening of the alimentary limb and common channel. Long-term vitamin deficiency sequelae potentially include osteoporosis and osteopenia (calcium and vitamin D metabolism), nephrolithiasis, night vision loss (vitamin A), and iron deficiency anemia.11
The potential side effects of steatorrhea, malodorous gas, and diarrhea are typically overstated. In comparative studies for DS and gastric bypass, the average number of bowel movements reported after switch averaged 1.68 daily for DS versus 1.18 daily for bypass.12 When present, diarrhea is linked most often to poor eating habits related to fatty foods, lactose intolerance, or bile salt diarrhea.
Non-nutrition-related late-term complications noted in our long term experience include adhesive bands with closed loop obstruction and potentially internal hernia.
Patient Selection
Careful patient selection is critical to minimizing adverse nutritional outcomes, particularly due to nonadherence. The rate of long-term follow-up in bariatric patients, in general terms, trends to less than 30 percent.13 Transition to a hypoabsorptive procedure carries risk of malabsorption of fat-soluble vitamins, minerals, potassium, and protein. Consequently, choosing this operation demands a high level of adherence to clinical follow-up, serial serum lab testing, and nutritional counseling. Repeated efforts to educate and reinforce the importance of supplementation and follow-up, a dependable system of communication, and a formal contract with patients are critical to assure long term follow-up and track nutritional progress.
Financially, malabsorptive operations require a lifelong modest monthly expense for supplements and vitamins. Generally, if patients lack the intellectual capacity to comprehend the regimen, its significance, or struggle to meet the financial responsibilities for supplements, candidacy for malabsorption should be carefully reconsidered.
Nutritional Guidelines
We follow or exceed the American Society for Metabolic and Bariatric Surgery (ASMBS) published guidelines for nutritional supplementation specific to DS.14 Patients who undergo GBDS undertake a standardized malabsorption surgery vitamin supplement regimen. The schedule requires bariatric formulated multivitamins with A, D, E, and K (ADEK MVI), two capsules taken twice daily, four calcium citrate 500mg chewable divided 4x daily, vitamin D 5000 IU tablet daily, and ferrous fumarate 60mg elemental daily.
The ADEK MVI (4 capsules) contains vitamin A (68% as beta carotene) 25,000 IU, vitamin D 5000 IU, vitamin E (as d-alpha-tocopherol acetate) 200mg, and vitamin K (as phylloquinone) 650mcg as well as vitamin C 180mg, thiamin 12mg, riboflavin 12mg, niacin (as niacinamide) 40mg, vitamin B6 (as pyridoxine) 12mg, folic acid 800mcg, vitamin B12 1000mcg, and Biotin 600mcg. Minerals include magnesium 200mg, zinc 45mg, selenium 140mcg, copper 4.5mg, and other trace elements.
We adjust each vitamin regimen for each patient based on serum lab values obtained every three months in the first year, every six months in the second year, and annually thereafter (if vitamin regimen and lab values remain stable). Our schedule for nutritional testing is a follows: vitamins A, D, E, K, comprehensive metabolic panel, complete blood count (CBC), ferritin, vitamin B12, folate, thiamine, hemoglobin A1c (HgbA1c), parathyroid hormone (PTH), copper, selenium, and zinc.
Path to Adoption
Incorporating GBDS conversion into practice is complex. Understanding that fewer than one percent of practices perform DS only adds to the challenge.2 Adoption requires establishing the nutritional infrastructure to support a malabsorption patient base, more frequent structured follow-up, dieticians trained in fat soluble vitamin management, and a programmatic commitment to long-term care.
The technical component involves learning BPD-DS, or related variants such as single anastomosis DS. The laparoscopic conversion of gastric bypass to DS is a challenging operation, demanding lysis of adhesions, small bowel resection, gastric reconstruction with gastrogastrostomy and partial gastrectomy, duodenal dissection and division, three small bowel anastomoses, and three potential mesenteric closures. Typically, the initial operations last upwards of 3 to 4 hours in inexperienced hands.
We recommend proctoring to learn the technical challenges and establish a mentoring relationship with an experienced surgeon and dietician. In terms of patient selection for the early learning curve, favorable qualities include younger patients, prior laparoscopic gastric bypass (known surgeon preferred), lower BMI, and female sex. This allows for a more measured introduction to the otherwise complex conversion operation. Staging the operation after the gastrogastrostomy or loop duodenal switch are also options.
Conclusion
Conversion from gastric bypass to DS is currently the most effective operation for disease recidivism after gastric bypass. Compared to revision based alternatives, DS offers the greatest efficacy for weight loss and comorbidity resolution with acceptable morbidity risks. Concerns for the technical complexity of the procedure, questions around patient adherence and nutritional risk, and the demands for lifelong support make GBDS conversion for many a stern programmatic challenge. Moreover, few bariatric programs nationally are structured to support DS in terms of committed registered dietitians or experienced surgeons. To advance access and address the growing need for conversion, more surgeons will need to accept the challenge of building the technical skill set and cultivating a broader bariatric program inclusive of malabsorptive procedures.
References
- Tran DD, Nwokeabia ED, Purnell S, et al. Revision of Roux-en-Y gastric bypass for weight regain: a systematic review of techniques and outcomes. Obes Surg. 2016;26:1627.
- English WJ, DeMaria EJ, Hutter MM, et al. American Society for Metabolic and Bariatric Surgery 2018 estimate of metabolic and bariatric procedures performed in the United States. Surg Obes Relat Dis. 2020;16(4):457–463.
- Ng PC, Sharp LS, Bermudez DM. Duodenal switch: fully stapled technique. Surg Obes Relat Dis. 2019;15(3):512.
- Ng P, Bermudez D, Sharp L, et al. Linear stapled duodenoileostomy for duodenal switch: short term retrospective analysis of 517 consecutive patients. Surg Obes Relat Dis. 2017;13(10):S190–S191.
- Keshishian A, Zahriya K, Hartoonian T, Ayagian C. Duodenal switch is a safe operation for patients who have failed other bariatric operations. Obes Surg. 2004;14:1187–1192.
- Parikh M, Pomp A, Gagner M. Laparoscopic conversion of failed gastric bypass to duodenal switch: technical considerations and preliminary outcomes. Surg Obes Relat Dis. 2007;3(6):611–618.
- Surve A, Zaveri H, Cottam D, et al. A mid-term outcomes of gastric bypass weight loss failure to duodenal switch. Surg Obes Relat Dis. 2016;12(9):1663–1670.
- Halawani HM, Bonanni F, Betancourt A, et al. Conversion of failed Roux-en-Y gastric bypass to biliopancreatic diversion with duodenal switch: outcomes of 9 case series. Surg Obes Relat Dis. 2017;13(8):1272–1277.
- Moon, RC, Alkhairi, L, Wier, A. et al. Conversions of Roux-en-Y gastric bypass to duodenal switch (SADI-S and BPD-DS) for weight regain. Surg Endosc. 2019. Online ahead of print.
- Cerfolio RJ, Bryant A, Canon C, et al. Is botulinum toxin injection of the pylorus during Ivor Lewis [corrected] esophagogastrectomy the optimal drainage strategy? Thorac Cardiovasc Surg. 2009;137(3):565–572.
- Lupoli R, Lembo E, Saldalamacchia G, et al. Bariatric surgery and long-term nutritional issues. World J Diabetes. 2017;8(11):464–474.
- Wasserberg N, Hamoui N, Petrone P, et al. Bowel habits after gastric bypass versus the duodenal switch operation. Obes Surg. 2008;18(12):1563–1566.
- Thereaux J, Lesuffleur T, Païta M, et al. Long-term follow-up after bariatric surgery in a national cohort. Br J Surg. 2017;104(10):1362–1371.
- Parrott J, Frank L, Rabena R, et al. American Society for Metabolic and Bariatric Surgery Integrated Health Nutritional Guidelines for the Surgical Weight Loss Patient 2016 update: micronutrients. Surg Obes Relat Dis. 2017;13(5):727–741.
Category: Past Articles, Review




