COVID-19 and Obesity—Nursing Accredited
 This is a complimentary continuing education activity for nurses. Nurses earn 1.0 Contact Hour.
This is a complimentary continuing education activity for nurses. Nurses earn 1.0 Contact Hour.
Course Instructions
- To access post-test and obtain credit, visit www.tinyurl.com/BTCE-August2021 (This activity is approved for nurses only).
- Participants must read the designated article and take, submit, and pass the post-test by August 1, 2022.
- Participants must answer at least 70% of the post-test questions correctly to pass and receive credit for the activity.
- Upon successful completion of the post-test, participants will receive a PDF certificate.
Department Overview
This department is dedicated to providing peer-reviewed, evidenced-based continuing education activities for integrated health clinicians who care for patients with overweight or obesity and their related comorbidities.
Course Overview
This educational program is designed to educate, through independent study, multidisciplinary clinicians who care for pre- and postoperative bariatric surgical patients on the impact of COVID-19 in individuals with severe obesity.
Course Description
This continuing education course is designed to educate, through independent study, integrated healthcare clinicians who care for the metabolic and bariatric surgical patient population.
Course Objectives
Upon completion of reading this article the participant should be able to:
- Define COVID-19, SARS CoV-2.
- Discuss obesity as a risk factor for severe COVID-19.
- Explain the etiology of COVID-19 infection on humans.
- Describe the obesity pandemic in the US.
- Explain how obesity affects individual’s immune system.
Target Audience
This accredited program is intended for nurses who treat bariatric and metabolic surgery patients.
Completion Time
This educational activity is accredited for a total of 1.0 contact hour (Nursing).
Author Disclosures
The authors have no conflicts of interest relevant to the content of this article.
Accreditation Summary
This educational program is provided by Matrix Medical Communications. Provider approved by the California Board of Registered Nursing, Provider Number 14887. Activity approved for 1.0 contact hour.
Provider Contact Information
Amanda Tolvaisa, Matrix Medical Communications, 1595 Paoli Pike, Suite 201, West Chester, PA 19380; Email: atolvaisa@matrixmedcom.com
A Message from the Department Editor
Tracy Martinez, RN, BSN, CBN
Ms. Martinez is Program Director of Wittgrove Bariatric Center in Del Mar, California.
Dear Colleagues:
I hope this finds you well in an unprecedented time in our lives and careers. COVID-19 has impacted so many aspects of our lives. I appreciate this timely article that discusses the impact of COVID-19 on our patient population and the increased risk of severe COVID-19 infections. It explains the etiology and pathophysiology of this aggressive virus on individuals who suffer from severe obesity.
Sincerely,
Tracy Martinez, RN, BSN, CBN
by Chowdhury S. Ferdous, MD, Dip. ABOM, CNSC, and Alexandra Wells, MD
Dr. Ferdous is Associate Physician, Geisinger Medical Center, in Danville, Pennsylvania. Dr. Wells is with Riverside Methodist Hospital, Department of Medical Education, in Columbus, Ohio.
Funding: No funding was provided.
Disclosures: The authors have no conflicts of interest relevant to the content of this article.
Abstract
Introduction: COVID-19 is a new pathogen without preexisting immunity in humans. Obesity is one of the major modifiable risk factors that affect COVID-19 adversely. We focused on association of obesity among patients infected with COVID-19, mechanism of severity, and possible intervention strategy.
Methods: Using keywords, “COVID-19” + “Obesity,“ only in abstract, in PubMed and Google Scholar, peer-reviewed papers were selected. Manuscripts that lacked data on obesity were excluded. A total of 46 abstracts were reviewed.
Conclusion: In summary, it has been identified by different bodies that obesity is a risk factor for poorer outcome in COVID-19 disease. We briefly discussed the mechanism that suggests underlying oxidative stress acts as the main driver for poorer outcome by causing pro-inflammatory state (inflammatory storm), driven by activation of CD-14 and CD-61 inflammatory monocytes producing larger amounts of IL-6 and other pro-inflammatory factors. Viral factors are not modifiable, but we can change the host factors to reduce severity of illness for patients with obesity. We briefly discussed, management of COVID-19 infection associated with obesity, based on the Obesity Medicine Association obesity management algorithm.
Keywords: COVID-19, coronavirus, obesity, inflammation, pandemic
Bariatric Times. 2021;18(8):10–14.
COVID-19 as a Pandemic
On December 30, 2019, a patient in Wuhan Jinyintan Hospital was ill from a pneumonia of unknown etiology. Due to surveillance protocols that had been established during the severe acute respiratory syndrome (SARS) outbreak of 2002–2003, three bronchoalveolar lavage (BAL) samples were collected.1 Real-time polymerase chain reaction (RT-PCR) assays on these samples were positive for pan-betacoronavirus, which was later identified as COVID-19, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). As the virus began to spread, the World Health Organization (WHO) declared the novel coronavirus outbreak a pandemic on March 11, 2020.2 As of March 6, 2021, in the United States (US), total cases were 28,714,163 and total deaths were 521,294.3
Since the first case was identified in the US on January 24, 2020, researchers have been active in identifying the characteristics of this disease. As COVID-19 is a newly identified pathogen, there is no known pre-existing immunity in humans.4–6 Based on the epidemiologic characteristics observed so far in China, everyone is assumed to be susceptible, although some might be more so than others.7 Individuals with comorbid conditions, such as hypertension, obesity, diabetes, cardiovascular disease, chronic respiratory diseases, cancer, and age over 60 years are at increased risk for infection and adverse outcomes, including mortality.4, 8,9
Obesity as a Pandemic
Obesity is a common, serious, and costly disease. From 1999–2000 through 2017–2018, the prevalence of obesity increased from 30.5 percent to 42.4 percent, and the prevalence of severe obesity doubled, increasing from 4.7 percent to 9.2 percent.10 The risk of death among patients with obesity is 2-to-3-times higher than individuals without obesity, and individuals with severe obesity have a 5-to-20-year decrease in life expectancy in comparison to individuals without obesity, when matched for age and gender.11,12 Obesity-related conditions include heart disease, stroke, Type 2 diabetes, and certain types of cancer that are some of the leading causes of preventable, premature death.13 The estimated annual medical cost of obesity in the US was $147 billion in 2008 US dollars; the annual medical cost for people who have obesity was $1,429 higher than those of normal weight.
Obesity, defined as a body mass index (BMI) between 30kg/m2 and <40kg/m2 or severe obesity, BMI of 40kg/m2 or above, increases risk of severe illness from COVID-19.
The use of lockdowns to combat the COVID-19 pandemic seems to have been successful from an epidemiological perspective, but it might have serious negative consequences on other health metrics.14 Lockdown seems to have affected metabolic health via combined deterioration of socioeconomic conditions, psychological security, and metabolic processes. To this end, approaches designed to contain the spread of COVID-19 might promote obesity and associated metabolic diseases.
The current pandemic has affected nearly every part of society, transforming our daily habits, lifestyle, work, family, and social cultures. Thus, we are seeing a collision effect of two ongoing pandemics.
Obesity as a Risk Factor for Severe COVID-19
Patients with obesity are more vulnerable to contracting COVID-19.15 Obesity is associated with a nearly threefold increased risk for severe COVID-19 with a dose-effect relationship between increasing BMI and the proportion of patients with severe illness.16 There was a 46 percent increased risk for testing positive17 and a 113 percent increased risk for hospitalization among patients with obesity. The studies also showed a 74 percent increased risk for intensive care unit (ICU) admission and a 66 percent increased risk for use of invasive mechanical ventilation (IMV) in patients with both obesity and COVID-19. Overall, there was a 48 percent increased risk of mortality associated with obesity in patients who contracted COVID-19.
The morbidity and mortality risk associated with obesity and COVID-19 is particularly relevant in the US because the prevalence of obesity is approximately 40 percent, versus a prevalence of 6.2 percent in China, 20 percent in Italy, and 24 percent in Spain.18 Reports from the US also showed that among patients with COVID-19 who are younger than 60 years of age, those with a BMI 30–35kg/m2 and over 35kg/m2 were 1.8 and 3.6 times more likely to be admitted to ICU, respectively, compared to those with a BMI lower than 30kg/m2.19
While the prevalence of obesity is lower in other countries, the association with severe COVID-19 infection remains. In the United Kingdom (UK), the NHS Intensive Care National Audit & Research Centre (ICNARC) report declared that 38 percent of patients admitted to ICU associated with COVID-19 had obesity.16 A retrospective study conducted in France showed that 76 percent of patients admitted to ICU for COVID-19 were at least overweight.20 Researches from China also showed that the presence of obesity increases the risk of severe COVID-19 approximately threefold, resulting in a longer hospital stay.21
Pathophysiology of COVID-19 Infection
Mechanism of COVID-19 infection. SARS-CoV-2 is one of many coronaviruses found in bats. In 2012, SARS-CoV-1 spread from bats to civets, and then to humans. Middle East respiratory syndrome (MERS) spread from bats to camels to humans. SARS-CoV-2 is thought to have spread from bats to pangolins to humans, and subsequently from human to human.
The SARS-CoV-2 life cycle begins when the virus invades susceptible host cells. ACE-2 binds to the receptor-binding domain (RBD) of spike proteins (S) (Figure 1),22 allowing for fusion with the host cell membrane. Positive sense single-stranded ribonucleic acid (RNA) is released, partially translated into SARS-CoV-2 polymerase protein, and transcribed. The resultant sub genomic RNA translational S, M, and E proteins are taken to the host cell endoplasmic reticulum and combined with nucleocapsid protein (N). Post Golgi processing, all elements are incorporated into a mature virion and traffic to the cell membrane for exocytosis of newly budded SARS-CoV-2 particles.
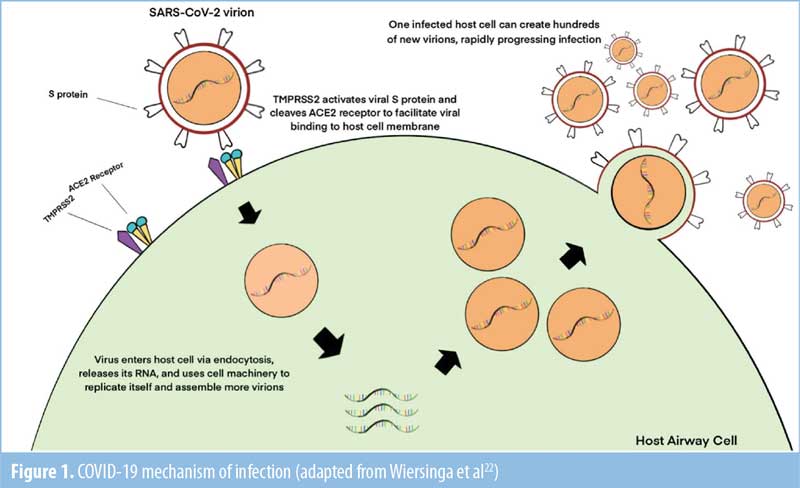
The ACE-2 receptor is used by the SARS-CoV-2 spike protein as a coreceptor for host cell entry.23 ACE-2 is highly expressed in heart and lung tissues. The expression of ACE-2 is even higher in adipose tissue than it is in the lungs, which is the main target tissue of COVID-19.24 Patients with obesity have an increased amount of adipose tissue and thus an increased number of ACE-2 expressing cells and ACE-2 receptors. This might explain the increased risk that patients with obesity have for contracting COVID-19.15
Pathophysiology of Disease Severity in COVID-19, Associated with Obesity
Presence of oxidative stress in obesity. Previous studies have shown that COVID-19 infection can induce the body to produce free radicals, leading to oxidative stress (OS). The production of cytokines and adipocytokines have proinflammatory downstream effects, allowing increased viral replication. The excessive free radicals can damage cell membranes and proteins, leading to organ damage, frequently seen in the lungs, heart, liver, and kidneys.25
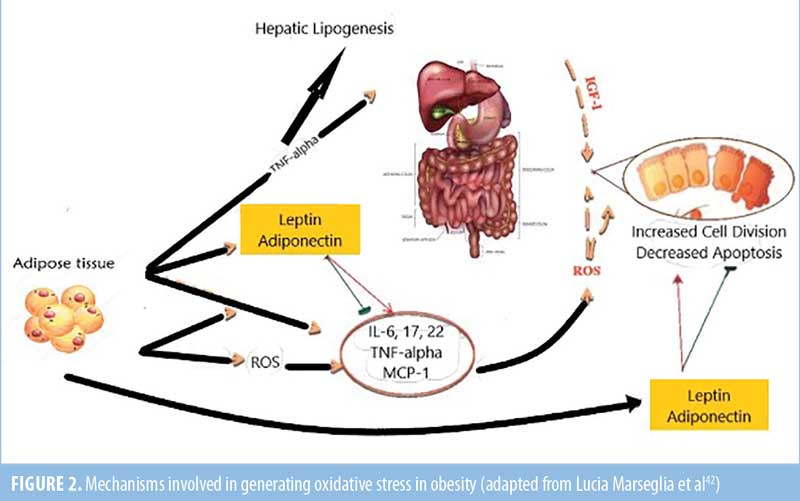
Preexisting hypercytokinemia in obesity. Since the discovery of leptin and adiponectin in the 1990s, adipose tissue has been recognized as an endocrine organ.26 Adipose tissue can also produce adipocytokines, like visfatin, which has been shown to activate CD14+ monocytes and NFκB, leading to an ongoing proinflammatory state.25 Due to this, patients with obesity live in a more inflammatory state than those without obesity.
Obesity-induced severe illness among patients with COVID-19 is promoted by the activation of CD14+ and CD16+ monocytes, which produce a large amount of interleukin (IL)-6 and other proinflammatory factors. IL-6 appears to be a key proinflammatory factor that triggers the cytokine “storm” associated with severe illness.27 Additionally, in patients with nonalcoholic fatty liver disease (NAFLD) and obesity, increased inflammatory activity in the liver and visceral fat are independently correlated with increased levels of IL-6.28 Taken together, these findings are suggestive of an additive/synergistic role in promoting greater severity of COVID-19 in patients with obesity.
Underlying preexisting comorbid conditions. Obesity-related comorbidities are related to the development of severe COVID-19 disease. Obesity is associated with an increased risk of diabetes mellitus, cardiovascular disease, and kidney disease, comorbidities that result in increased vulnerability to pneumonia-associated organ failures. Even in the absence of known comorbidities of obesity, the presence of hypertension, dyslipidemia, prediabetes, and insulin resistance, referred to as metabolic syndrome, predisposes individuals to cardiovascular events, atherosclerosis, and impaired immune response. Metabolic syndrome alone is related to a twofold increase in cardiovascular events and a 1.5-fold increase in all-cause mortality.29 Many studies thus far have shown that comorbidities, like cardiovascular disease, hypertension, diabetes mellitus, and chronic lung disease, are associated with severe clinical course of COVID-19 and increased mortality.7,30 Studies using patients from UK Biobank showed increased liver adiposity correlated with more severe COVID-19 disease.31 It is hypothesized that the subclinical inflammation and increased levels of IL-6 associated with NAFLD might increase susceptibility to COVID-19.
Pulmonary dysfunction in obesity. Patients with obesity often have respiratory dysfunction, which can manifest in the upper airways, lower airways, or lung parenchyma. They are predisposed to developing restrictive lung disease and obesity hypoventilation syndrome, which are characterized by alterations in respiratory mechanisms, increased airway resistance, impaired gas exchange, low lung volumes, and decreased muscle strength. These individuals, subsequently, are more susceptible to hypoventilation-associated pneumonia, pulmonary hypertension, and cardiac stress.
Cardiac dysfunction in obesity. Hypertension is 3-to-5-times more common in patients with obesity, in comparison to individuals within the ideal weight range.11 All components of metabolic syndrome are independent predictors of cardiovascular events, such as stroke, cardiomyopathy, coronary artery disease, myocardial infarction, congestive heart failure, and sudden cardiac death.32 Central or visceral obesity is highly correlated to the risk of cardiovascular disease.12,33,34 The left atrium is important in all phases of the cardiac cycle. Enlargement of the atria is associated with obesity and has been also linked with an increased risk of mortality.35 Cardiac and endothelial dysfunction increase the risk of severe COVID-19 and mortality among patients with obesity.
Endothelial dysfunction in obesity. The endothelium modulates vascular function by production of nitric oxide (NO), which protects the vasculature against the development of atherosclerosis and thrombosis. Visceral fat reduces NO availability, leading to endothelial dysfunction. In a healthy state, perivascular adipose tissue (PVAT) secretes factors that influence vasodilation by increasing NO availability. This effect is lost in patients with obesity, leading to endothelin 1/NO imbalance. Thus, in patients with obesity, vasculature not only represents a main target of PVAT-derived pro-inflammatory cytokines, but it is also considered an important source of low-grade inflammation and oxidative stress, which contributes to endothelial dysfunction among patients with obesity. This preexisting endothelial dysfunction increases thrombotic complications among patients with obesity.36
Immune system dysfunction in obesity.Obesity is a state of chronic, low-grade inflammation. This affects patients’ innate and adaptive immune response.37 Pathological adipocyte hypertrophy38 contributes to mechanical/sheer stress on the extracellular environment.39 It also leads to lipolysis, increased circulating free fatty acids (FFAs), and activation of the endoplasmic reticulum stress response. FFAs are known to increase insulin resistance and activate inflammatory pathways.20 Under these conditions, lower concentrations of adiponectin (an anti-inflammatory adipokine),39 and higher concentrations of leptin (a pro-inflammatory adipokine) are seen.40
The chronic inflammation seen in obesity leads to macrophage dysfunction in the setting of infection.41 Obesity also impairs the memory CD8+ T-cell response as evident in influenzae virus infection, resulting in increased mortality, elevated viral titers, and worse lung pathology.42
Clinical Aspect of Severe COVID-19 Disease Among Patients with Obesity
Taking care of patients with obesity has always presented numerous challenges to healthcare staff. Patients with obesity are more difficult to intubate, can be difficult to examine, might require special imaging and hospital beds, and present challenges for nursing staff with positioning and transport. Due to their altered respiratory physiology, patients with obesity require higher positive end expiratory pressure (PEEP). When a patient with obesity has poor oxygenation or ventilation, they might respond well to prone positioning and extra-corporeal support.44
Social Stigma Associated with Obesity and COVID-19
There is a pervasive and strong culture of weight stigma around the world. Weight stigma is likely to drive weight gain and poor health, and it is particularly prevalent and detrimental in healthcare settings, with documented high levels of “anti-fat“ bias in healthcare providers. Patients with obesity also tend to be socially isolated.
COVID-19 has led to stigma and discrimination among various groups of people in different populations.45 As COVID-19 is an airborne disease, both social coercion and social isolation play a role in patients hiding symptoms to avoid discrimination, thereby delaying diagnosis and treatment. Stigmas, like those associated with obesity and COVID-19 infection, can undermine the social matrix of the country and pose as barriers for disease control.
Patients with obesity who become infected with COVID-19 are likely to suffer from an additive effect of the stigmas from these two pandemics, leading to significant delay in detection of the early phase of the disease and early management.
Management of COVID-19 and Obesity
Nutritional therapy. Nutritional therapy as first-line therapy of COVID-19 infection should be implemented in standard practice regardless of patients’ BMI. Anti-inflammatory effects of different dietary components, like proteins, omega-3 fatty acids, vitamin A, vitamin C, dietary fiber, selenium, and copper have anti-inflammatory effects. Polar lipids have anti-thrombotic effects. Vitamin E, iron, and zinc support immune function. Vitamin A, vitamin C, and vitamin D protect against respiratory infection. Additionally, omega-3 fatty acids and vitamins A, C, and E all function as antioxidants, reducing free radicals. These micronutrients might be beneficial in decreasing the severity of COVID-19 disease and enhancing recovery. Optimizing micronutrient status has the potential to improve clinical outcomes and decrease hospital stays. It is worth mentioning that to reduce the risks and consequences of infection, the intake for some micronutrients might exceed the recommended dietary allowances, as infection and other stressors can reduce micronutrient availibility.46
Obesity is associated with changes in intestinal flora.47,48 Changes in gut microbiota has been linked to all obesity-related comorbidities. Dysregulation of gut microbiota could also be associated with changes in the immune system and thus more severe COVID-19 infection. It is proposed that changes in the gut microbiota composition through diet can aid in healing and recovery from COVID-19 infection.49 The use of prebiotics and probiotics to restore and balance intestinal flora could be an effective treatment to reduce severity of bacterial and viral infections.50 The effect of a high-fiber diet and/or probiotics on the outcome of SAR-CoV-2 infection is yet to be investigated.
Physical exercise. Various safety measures taken worldwide by authorities to reduce transmission of COVID-19 infection likely led to reduced physical activity in the general population. Physical exercise is of utmost importance in weight loss and decreasing insulin resistance. Patients should be given recommendations on how to increase physical activity during the times of social isolation using evidence-based exercise programs. There is a dose-dependent relationship between physical activity and major health outcomes; increasing daily step counts has been shown to improve mortality and decrease cardiovascular risk.51 This can be achieved by breaking sitting time with two minutes of walking every 20 to 30 minutes.52 Taking the stairs and adding short walks can be implemented in nearly any mobile patient’s daily routine. Considering the current social distancing guidelines, an exercise regime performed as intermittent high intensity interval training (I-HITT), incorporating body weight exercises and using large muscle groups, would be ideal to induce positive adaptations in both the cardiorespiratory system and the skeletal muscle. Choice of exercise could include jogging in place, burpees, rope skipping, mountain climbers, air-squat, jumping lunges, push-ups, and single step climbing.53
Pharmacotherapy and vaccination. Early use of pharmacotherapy for weight loss during this pandemic is more important than ever. Providers should continue to counsel patients to take their medication as prescribed, including ACEIs and ARBs, reminding them to keep at least a 30-day supply of medication available at all times. Obesity could be linked to lower vaccine responses for numerous diseases, such as influenza,54 Hepatitis B,55–57 and tetanus.58 This is likely due to altered dysfunctional innate and adaptive immunity. It is important to keep in mind that COVID-19 vaccination might not be as protective among patients with obesity as it is in those without obesity.59
Stress management. Individuals enrolled in a weight-loss program experienced higher levels of stress during the COVID-19 pandemic compared to the pre-pandemic era due to various mental health challenges originating from changes in employment status, having school-aged children at home along with them, and other disruptions of daily routines.59 Higher stress levels were associated with greater levels of anxiety, which in turn were associated with less time for weight management efforts among individuals for whom weight loss was recommended.
Weight management specialists should focus on educating patients in programs regarding coping with stress, providing helpful information on resources, using online platforms, and keeping them up to date by using frequent telemedicine or telephone visits.
Conclusion
Disparities in healthcare are impacted by COVID-19 and obesity, which are both pandemics in their own right. Stay-at-home orders and limited activity can negatively impact obesity. The purpose of this review is to help providers, patients, and associates who are involved in weight management to better understand the underlying mechanisms of severe COVID-19 among patients with obesity, as well as areas for further research and potential intervention, during a time when our understanding of this disease is rapidly evolving.
References
- WHO. Report of the WHO-China Joint Mission on Coronavirus Disease 2019 (COVID-19). https://www.who.int/docs/default-source/coronaviruse/who-china-joint-mission-on-covid-19-final-report.pdf. 2020.
- Cucinotta D, Vanelli M. WHO declares COVID-19 a pandemic. Acta Biomed. 2020;91:157–160.
- CDC. United States COVID-19 cases, deaths, and laboratory testing (NAATs) by state, territory, and jurisdiction. https://covid.cdc.gov/covid-data-tracker/#cases_casesper100klast7days.
- Team CC-R. Preliminary estimates of the prevalence of selected underlying health conditions among patients with coronavirus disease 2019: United States, February 12–March 28, 2020. Morb Mortal Wkly Rep. 2020;69:382–386.
- Hu B, Guo H, Zhou P, Shi ZL. Characteristics of SARS-CoV-2 and COVID-19. Nat Rev Microbiol. 2021;19(3):141–154.
- Muralidar S, Ambi SV, Sekaran S, Krishnan UM. The emergence of COVID-19 as a global pandemic: understanding the epidemiology, immune response and potential therapeutic targets of SARS-CoV-2. Biochimie. 2020;179:85–100.
- Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72,314 cases from the Chinese Center for Disease Control and Prevention. JAMA. 2020;323:1239–1242.
- Chu Y, Yang J, Shi J, et al. Obesity is associated with increased severity of disease in COVID-19 pneumonia: a systematic review and meta-analysis. Eur J Med Res. 2020;25:64.
- Kim SR, Nam SH, Kim YR. Risk factors on the progression to clinical outcomes of COVID-19 patients in South Korea: using national data. Int J Environ Res Public Health. 2020;17.
- Hales CM, Carroll MD, Fryar CD, Ogden CL. Prevalence of obesity and severe obesity among adults: United States, 2017–2018. NCHS Data Brief. 2020:1–8.
- Cannon CP. Obesity-related cardiometabolic complications. Clin Cornerstone. 2008;9:11–19; discussion 20–22.
- Cannon CP. Cardiovascular disease and modifiable cardiometabolic risk factors. Clin Cornerstone. 2008;9:24–38.
- Executive Summary: Guidelines (2013) for the Management of Overweight and Obesity in Adults. Obesity. 2014;22(suppl 2).
- Clemmensen C, Petersen MB, Sorensen TIA. Will the COVID-19 pandemic worsen the obesity epidemic? Nat Rev Endocrinol. 2020;16:469–470.
- Albashir AAD. The potential impacts of obesity on COVID-19. Clin Med (Lond). 2020;20:e109–e113.
- Gao F, Zheng KI, Wang XB, et al. Obesity is a risk factor for greater COVID-19 severity. Diabetes Care. 2020;43:e72–e74.
- Popkin BM, Du S, Green WD, et al. Individuals with obesity and COVID-19: a global perspective on the epidemiology and biological relationships. Obes Rev. 2020;21:e13128.
- World Health Organization. Prevalence of obesity among adults, BMI≥30, age-standardized – estimates by country. 2017. https://apps.who.int/gho/data/view.main.CTRY2450A?lang=en. Accessed July 22, 2021.
- Simonnet A, Chetboun M, Poissy J, et al. High prevalence of obesity in severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) requiring invasive mechanical ventilation. Obesity (Silver Spring). 2020;28:1195–1199.
- Guilherme A, Virbasius JV, Puri V, Czech MP. Adipocyte dysfunctions linking obesity to insulin resistance and Type 2 diabetes. Nat Rev Mol Cell Biol. 2008;9:367–377.
- Hotamisligil GS. Inflammation and metabolic disorders. Nature. 2006;444:860–867.
- Wiersinga WJ, Rhodes A, Cheng AC, et al. Pathophysiology, transmission, diagnosis, and treatment of Coronavirus Disease 2019 (COVID-19): a review. JAMA. 2020;324(8):782–793.
- Hoffmann M, Kleine-Weber H, Schroeder S, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271–280 e8.
- Jia X YC, Lu S. Two things about COVID-19 might need attention. Preprints, February 23, 2020.
- Moschen AR, Kaser A, Enrich B, et al. Visfatin, an adipocytokine with proinflammatory and immunomodulating properties. J Immunol. 2007;178:1748–1758.
- Funcke JB, Scherer PE. Beyond adiponectin and leptin: adipose tissue-derived mediators of inter-organ communication. J Lipid Res. 2019;60(10):1648–1684.
- Feng G, Zheng KI, Yan QQ, et al. COVID-19 and liver dysfunction: current insights and emergent therapeutic strategies. J Clin Transl Hepatol. 2020;8:18–24.
- van der Poorten D, Milner KL, Hui J, et al. Visceral fat: a key mediator of steatohepatitis in metabolic liver disease. Hepatology. 2008;48:449–457.
- Mottillo S, Filion KB, Genest J, et al. The metabolic syndrome and cardiovascular risk a systematic review and meta-analysis. J Am Coll Cardiol. 2010;56(14):1113–1132
- Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–1062.
- Stonehill M. Obesity, fatty liver play role in increased risk for COVID-19. Healio. 2020. https://www.healio.com/news/hepatology/20200701/qa-obesity-fatty-liver-play-role-in-increased-risk-for-covid19
- Knudson JD, Dincer UD, Bratz IN, et al. Mechanisms of coronary dysfunction in obesity and insulin resistance. Microcirculation. 2007;14:317–338.
- Gustafson B. Adipose tissue, inflammation and atherosclerosis. J Atheroscler Thromb. 2010;17:332–341.
- Aronne LJ, Nelinson DS, Lillo JL. Obesity as a disease state: a new paradigm for diagnosis and treatment. Clin Cornerstone. 2009;9:9–25.
- Benjamin EJ, D’Agostino RB, Belanger AJ, et al. Left atrial size and the risk of stroke and death. The Framingham heart study. Circulation. 1995;92:835–841.
- Virdis A. Endothelial dysfunction in obesity: role of inflammation. High Blood Press Cardiovasc Prev. 2016;23:83–85.
- Andersen CJ, Murphy KE, Fernandez ML. Impact of obesity and metabolic syndrome on immunity. Adv Nutr. 2016;7:66–75.
- Naaz A, Holsberger DR, Iwamoto GA, et al. Loss of cyclin-dependent kinase inhibitors produces adipocyte hyperplasia and obesity. FASEB J. 2004;18:1925-1927.
- Deng J, Liu S, Zou L, et al. Lipolysis response to endoplasmic reticulum stress in adipose cells. J Biol Chem. 2012;287:6240–6249.
- Ouchi N, Parker JL, Lugus JJ, Walsh K. Adipokines in inflammation and metabolic disease. Nat Rev Immunol. 2011;11:85–97.
- Ahn SY, Sohn SH, Lee SY, et al. The effect of lipopolysaccharide-induced obesity and its chronic inflammation on influenza virus-related pathology. Environ Toxicol Pharmacol. 2015;40:924–930.
- Zimorovat A, Mohammadi M, Ramezani-Jolfaie N, Salehi-Abargouei A. The healthy Nordic diet for blood glucose control: a systematic review and meta-analysis of randomized controlled clinical trials. Acta Diabetol. 2020;57:1–12.
- Marseglia L, Manti S, D’Angelo G, et al. Oxidative stress in obesity: a critical component in human diseases. Int J Mol Sci. 2014;16:378–400.
- Parameswaran K, Todd DC, Soth M. Altered respiratory physiology in obesity. Can Respir J. 2006;13:203–210.
- Chopra KK, Arora VK. COVID-19 and social stigma: role of scientific community. Indian J Tuberc. 2020;67:284-285.
- Fernandez-Quintela A, Milton-Laskibar I, Trepiana J, et al. Key aspects in nutritional management of COVID-19 patients. J Clin Med. 2020;9.
- Sanz Y, Moya-Perez A. Microbiota, inflammation and obesity. Adv Exp Med Biol. 2014;817:291–317.
- Sanmiguel C, Gupta A, Mayer EA. Gut microbiome and obesity: a plausible explanation for obesity. Curr Obes Rep. 2015;4:250–261.
- Villapol S. Gastrointestinal symptoms associated with COVID-19: impact on the gut microbiome. Transl Res. 2020;226:57–69.
- Conte L, Toraldo DM. Targeting the gut-lung microbiota axis by means of a high-fibre diet and probiotics may have anti-inflammatory effects in COVID-19 infection. Ther Adv Respir Dis. 2020;14:1753466620937170.
- US Department of Health and Human Services. Physical activity guidelines for americans, 2nd edition. Washington, D.C. Department of Health and Human Services. 2018.
- Loh R, Stamatakis E, Folkerts D, et al. Effects of interrupting prolonged sitting with physical activity breaks on blood glucose, insulin and triacylglycerol measures: a systematic review and meta-analysis. Sports Med. 2020;50:295–330.
- Menz V, Marterer N, Amin SB, et al. Functional vs. running low-volume high-intensity interval training: effects on VO2max and muscular endurance. J Sports Sci Med. 2019;18:497–504.
- Neidich SD, Green WD, Rebeles J, et al. Increased risk of influenza among vaccinated adults who are obese. Int J Obes (Lond). 2017;41:1324–1330.
- Weber DJ, Rutala WA, Samsa GP, et al. Obesity as a predictor of poor antibody response to hepatitis B plasma vaccine. JAMA. 1985;254:3187–3189.
- Simo Minana J, Gaztambide Ganuza M, Fernandez Millan P, Pena Fernandez M. Hepatitis B vaccine immunoresponsiveness in adolescents: a revaccination proposal after primary vaccination. Vaccine. 1996;14:103–106.
- Young MD, Gooch WM III, Zuckerman AJ, et al. Comparison of a triple antigen and a single antigen recombinant vaccine for adult hepatitis B vaccination. J Med Virol. 2001;64:290–298.
- Eliakim A, Schwindt C, Zaldivar F, et al. Reduced tetanus antibody titers in overweight children. Autoimmunity. 2006;39:137–141.
- Pellegrini CA, Webster J, Hanh KR, et al. Relationship between stress and weight management behaviors during the COVID‐19 pandemic among those enrolled in an internet program. Obes Sci Pract. 2020;7(1):129–134.
Category: Integrated Health Continuing Education, Past Articles





Thank you for this opportunity to gain further education on obesity. I am a new Outcomes Manager for our site’s Bariatric Program. I appreciate any future education r/t obesity.